Aortic chondroid neoplasia in two Labrador Retriever dogs

Author information
Kohnken R., Durham J.A., Premanandan C., Scansen B.A. Aortic chondroid neoplasia in two Labrador Retriever dogs // J Vet Cardiol. 2015 Dec;17(4):314-20.
Abstract
In the same week, two Labrador Retriever dogs presented to The Ohio State University Veterinary Medical Center for cardiac evaluation. The presenting signs in both dogs included: weight loss, weakness, lethargy, and decreased femoral pulses. The first dog presented in cardiogenic shock and biventricular congestive heart failure, which initially responded to treatment; however, the dog was euthanized due to deteriorating clinical condition. In contrast, the second dog had a milder clinical course without signs of congestive heart failure, and remained stable over the 2-month period of clinical evaluation prior to euthanasia. Echocardiographic evaluation revealed a dilated cardiomyopathy phenotype in the first dog, while a space-occupying intraluminal mass originating at the aortic valve with preserved left ventricular systolic function was observed in the second dog. At autopsy, each dog had a large obstructive luminal mass affecting the ascending aorta and arch. Histopathology revealed that the mass in the first dog was consistent with a benign chondroma, while in the second dog the morphologic characteristics, mitotic activity, and infiltrative growth justified a diagnosis of chondrosarcoma. This report presents the contrasting clinical disease progression and findings in two dogs with aortic neoplasia, with a proposed pathogenesis of cardiac failure secondary to aortic neoplasia.
KEYWORDS: Canine; Chondroma/chondrosarcoma; Congestive heart failure; Dilated cardiomyopathy
Introduction
An approximately 10-year-old female, spayed Labrador Retriever (Dog 1) weighing 28.1 kg was presented to The Ohio State Veterinary Medical Center with a 2-week history of progressive weight loss, inappetence, cough, and dyspnea. The dog was tetraparetic with weak femoral pulses, a heart rate of 140 beats per minute, and a respiratory rate of 44 per minute. On auscultation, there were increased bronchovesicular sounds on the left and right sides of the thorax with no cardiac murmur detected. Arterial blood pressure measured 70 mmHg by indirect Doppler measurement from the left hind limb. Complete blood count and serum biochemistry results were within reference intervals. Thoracic radiographs revealed rounding of the cardiac silhouette with predominately leftsided chamber enlargement, and a diffuse and patchy interstitial-to-alveolar pattern throughout the lungs, as well as mild pleural effusion, which were compatible with biventricular congestive heart failure. An echocardiogram was performed, which revealed moderate left ventricular dilatation, impaired left ventricular systolic function, and left atrial enlargement — consistent with a dilated cardiomyopathy phenotype (left ventricular internal dimension in diastole of 54.5 mm; left ventricular internal dimension in systole of 49.5 mm; linear percent fractional shortening of 9.2%; and left atrial diameter from a right parasternal long axis view of 52.4 mm; Fig. 1, Video 1).
Retrospectively, an abnormal soft tissue echogenicity was apparent in the aortic arch, but was not appreciated at the time of initial evaluation.
The dog was hospitalized and received furo- semide 60 mg IV q 12 h, and positive inotropes including pimobendan at 10 mg PO q 12 h, and dobutamine as a continuous IV infusion at 3—6 mcg/kg/min to maintain a systolic arterial pressure of >100 mmHg. The respiratory rate normalized within 24 h and the dog was more willing to ambulate. The dog then began to vomit repeatedly, so ondansetron 6 mg IV q 8 h, and maropitant citrate 30 mg SQ q 24 h were administered. Repeat thoracic radiographs showed resolution of the pleural effusion with a mild, resolving interstitial pattern in the right caudal lung lobe. The dog then developed diarrhea and repeat serum biochemistry showed moderate azotemia, with a serum urea nitrogen level of 52 mg/dL (reference interval 5—20 mg/dL) and a creatinine of 4.4 mg/dL (reference interval 0.6—1.6 mg/dL). However, the dog remained inappetent and lethargic. A complete abdominal ultrasound was performed, which revealed decreased caudal aortic blood flow without evidence of a thrombus or mass. Due to its deteriorating clinical condition, the dog was euthanized.
The following week, a 6.5-year-old male, castrated Labrador Retriever (Dog 2) weighing 28.6 kg presented to The Ohio State University Veterinary Medical Center with a 2-month history of collapse after exercise and intermittent, yet progressive, hind limb weakness that was worsened by activity. Physical examination documented an absent right femoral pulse, with mild bilateral hind limb weakness and muscle atrophy. Neurologic examination revealed mild hind limb ataxia, a decrease in hind limb conscious proprioception, mild hind limb weakness, and thoracolumbar pain. On thoracic auscultation, a Grade IV/VI left basilar systolic heart murmur with radiation to the right thorax was evident. Complete blood count and serum biochemistry results were unremarkable. Two-dimensional and Doppler echocardiography revealed a large, intraluminal, hyperechoic mass lesion extending from the non-coronary cusp of the aortic valve to the descending aorta, resulting in mild obstruction (peak left ventricular outflow velocity of 3.25 m/s, corresponding to a peak transaortic valve systolic pressure gradient of 45 mmHg) and mild aortic valve insufficiency (Fig. 2, Video 2). Left ventricular wall thickness and luminal dimensions were normal. Thoracic radiographs were unremarkable. Indirect systolic arterial blood pressure obtained by Doppler was normal in the right and left forelimbs (115 mmHg), decreased in the left hind limb (80 mmHg), and not attainable in the right hind limb. The dog was prescribed clopidogrel 75 mg PO q 12 h. Hemoptysis was noted 2 weeks after starting clopidogrel, at which time the packed cell volume was 55%, prothrombin time was 56 s (reference interval 12—17 s) and partial thromboplastin time was >250 s (reference interval 71—102 s). The clopi- dogrel was discontinued, and vitamin K was prescribed at 100 mg PO in the morning and 50 mg PO in the evening. Two weeks later the clotting times had normalized, with a prothrombin time of 8.1 s and a partial thromboplastin time of 11.7 s. A repeat echocardiogram 1 month after diagnosis revealed subjective enlargement of the aortic mass with greater involvement of the aortic valve leaflets. The chamber dimensions and estimates of left ventricular systolic function were unchanged. As the dog’s activity level remained poor and due to progressive growth of the mass, the dog was euthanized.
Figure 2 Echocardiographic images from the second case of this report: a 6.5-year-old male, castrated Labrador Retriever with hind limb weakness and a new- onset heart murmur
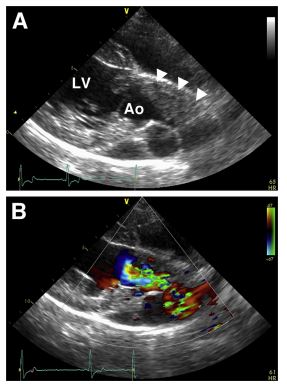
Panel A: Right parasternal long- axis view of the left ventricular outflow tract and aorta showing a hyperechoic intraluminal mass (arrowheads) within the ascending aorta. The mass appeared to be lateralized to the cranial aspect of the vessel. Based on short-axis echocardiographic views, invasion of right and non-coronary cusps of the aortic valve could be appreciated. Panel B: Same view as Figure 2A, with color Doppler imaging demonstrating turbulent blood flow around the intraluminal mass during systole. Ao, aorta; LV, left ventricle.
Figure 1 Echocardiographic images from the first case of this report: a 10-year-old female, spayed Labrador Retriever with biventricular congestive heart failure
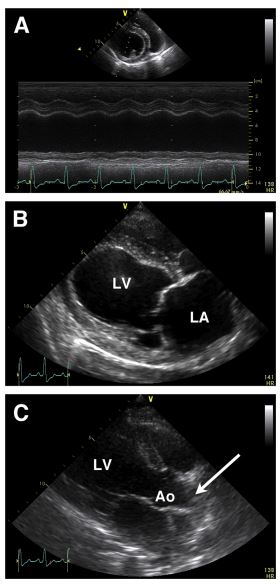
Panel A: Right parasternal short-axis M-mode image of the left ventricle just below the level of the mitral valve demonstrating left ventricular dilation and depressed systolic function, with hypokinesia of the left ventricular posterior wall. Panel B: Right parasternal long-axis four- chamber view demonstrating severe left ventricular dilatation and left atrial enlargement. Panel C: Right parasternal long-axis view of the left ventricular outflow tract and ascending aorta. A suspect hyperechoic, intraluminal mass can be appreciated within the ascending aorta (arrow), but was only noted in retrospect after the postmortem exam was completed. LV, left ventricle; LA, left atrium; Ao, aorta.
On postmortem examination of Dog 1, the heart was moderately enlarged with a rounded apex and it weighed 300 g (1% of body weight). The epi- cardial surface of both ventricles contained dozens of well-demarcated red foci (0.2—0.5 cm). The proximal aorta and aortic arch contained a tightly adherent intraluminal multi-nodular firm grey mass measuring 9.0 x 2.0 x 2.0 cm (Fig. 3A). On cut section, the mass was lobulated and waxy in consistency. A limited autopsy was performed to evaluate the heart only in Dog 2. Gross appearance of the aortic mass was similar to the previous case: a 5.0 x 3.0 x 2.0 cm multi-nodular firm tan mass partially occluded the proximal aorta. However, there was grossly evident invasion of the mass within the myocardium at the heart base, aortic valve annulus, and left ventricular wall (Fig. 3B).
Histologically, the aortic mass in Dog 1 was composed of poorly cellular lobules containing well-differentiated chondrocytes embedded in chondroid matrix (Fig. 4A and B). There were occasional binucleated cells and rare apoptotic cells; however, there were no mitoses seen. The lobules were circumferentially adherent to the aortic intima and there was a segmental separation of the aortic intima from the underlying fibroelastic tunic. There was a central lumen evident within the mass that was interpreted as degeneration and necrosis of the tumor tissue. Several subepicardial coronary arteries were characterized by acute fibrinoid necrosis with periarterial neutrophilic and lymphoplasmacytic inflammation. Within the subendocardium, there was multifocal degeneration and necrosis of myofibers.
Additionally, there were multifocal areas of chronic interstitial reactive fibrosis apparent throughout the ventricular myocardium. Histology of the lung revealed multiple large subpleural foci characterized by fibrosis, hemorrhage, and alveolar histiocytosis, consistent with chronic infarcts. Other systemic findings included severe hepatic nodular regeneration and interlobular fibrosis (cirrhosis), acute tubular necrosis within the renal cortex, focal segmental glomerulosclerosis, and chronic interstitial nephritis. In Dog 2, the luminal mass consisted of moderately cellular, poorly demarcated lobules containing moderately differentiated chondrocytes in a myxoid to chondroid matrix. Atypia was moderate, numerous mitoses were noted (23 per 10 400 x high power fields), and there was extensive necrosis of the tumor tissue. There was multifocal invasion of the mass into the aortic wall and surrounding myocardium (Fig. 4C and D).
Discussion
These cases provide examples of a rare diagnosis in any species: intra-aortic neoplasm. The neoplasms were morphologically chondroid in origin: the first was benign and the second was malignant. The diagnosis of chondroma was made for Dog 1, which was based on the benign histologic appearance and lack of invasion. This was in contrast to Dog 2, whose mass was grossly and histologically invasive into the surrounding tissues, with moderate cellular atypia warranting a diagnosis of chondrosarcoma. Once the signs developed, Dog 1 had a rapidly progressive clinical course due to a dilated cardiomyopathy phenotype and biventricular congestive heart failure. The obstruction from the aortic tumor is believed to have led to chronically elevated afterload, progressive myocardial fibrosis, and eventual myocardial dysfunction, as can occur with valvular aortic stenosis.1 This dysfunction then contributed to systemic lesions consistent with hypoxemia, including: coronary arterial necrosis, subendocardial myocardial necrosis, and acute tubular necrosis in the kidney. However, it is impossible to completely exclude an underlying primary cardiomyopathy in this dog. In contrast, Dog 2 had a histologically more-aggressive neoplasm but remained relatively stable upon diagnosis, with the only major clinical findings of an absent femoral pulse and hind limb weakness. It is unknown whether this dog would have also progressed to a dilated cardiomyopathy phenotype. While benign tumors are generally considered to be of lesser clinical concern than malignant tumors, the anatomic location may alter this outlook when the tumor compresses or obstructs vital structures such as the aorta.
Figure 3 Gross pathologic photographs of the Labrador Retriever dogs. Dog 1 (A) had a 9.0-cm firm, lobulated grey mass (arrow) adherent to the intimal surface of the aortic arch and ascending aorta
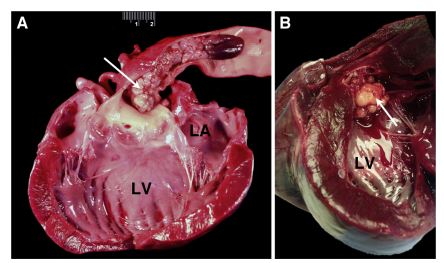
The left ventricle (LV) and left atrium (LA) were moderately dilated. Dog 2 (B) had a 5.0-cm firm, lobulated tan mass (arrow) involving the ascending aorta and aortic valve with invasion into the heart base.
Figure 4 Histologic images of the aortic tumors from the Labrador Retriever dogs
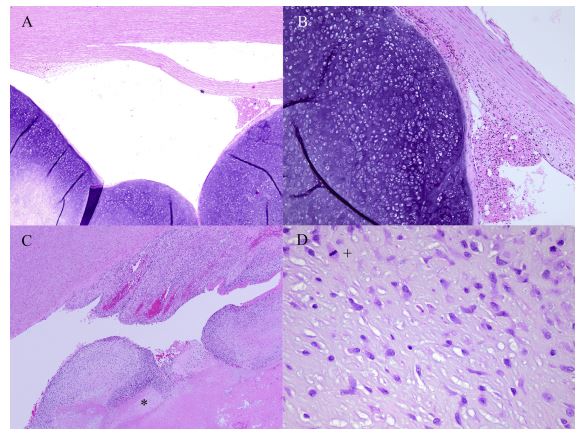
The mass from Dog 1 is composed of a poorly cellular and lobular neoplasm characterized by well-differentiated chondrocytes in a chondroid matrix (A). The mass is adherent to, but not invading, the aortic intima (B). The mass from the Dog 2 (C) can be seen to invade the myocardium (*) and is composed of a poorly cellular and lobular neoplasm characterized by moderately differentiated chondrocytes featuring moderate atypia and mitoses (+) (D). Hematoxylin and Eosin stain. Magnifications 40x (A); 100x (B), (C); 400x (D).
Primary tumors of the great vessels are rare in dogs, with few case reports available in the liter- ature.2-5 As of 2011, aortic chondrosarcoma had been reported in three dogs.2,4,6 A case of a 13- year-old Lhasa Apso had a similar presentation to those discussed here; it presented with tetraparesis, and was diagnosed postmortem with aortic chondrosarcoma affecting the aortic arch.2 The other two canine cases in the literature describe a clinical presentation of collapse due to an obstructive aortic arch mass resulting in aortic dissection and an aorto-pulmonary fistula,6 and acute pain with hind limb paresis due to acute occlusion of the abdominal aorta.4 Both human and canine reports of aortic neoplasia describe the difficulty of antemortem diagnosis due to variability of clinical presentation and pro- gression.2,4-10 Clinical signs may be related to thrombotic disease or reduced peripheral perfusion. Presenting complaints of hind limb lameness, pain, or paresis appear common in dogs.2,4 In both of the cases presented above, the femoral pulses were weak to absent. To the authors’ knowledge, there has previously not been a report of a dog presenting in cardiogenic shock and heart failure with a dilated cardiomyopathy phenotype secondary to an intraluminal aortic neoplasm.
In humans, clinical differential diagnoses for intra-aortic neoplasia predominantly include sarcomas, with the human medical literature reporting undifferentiated sarcoma most commonly followed by malignant fibrous histiocytoma, angiosarcoma, leiomyosarcoma, and others.3,10 In primary vascular neoplasms, malignancies are much more common than benign tumors.10 Secondary tumors, implying those that involve the aorta by invasion from nearby structures, are far more common than primary aortic neoplasms.10 Sarcomatous vascular tumors have been reported in venous and arterial systems in humans, with the aorta being the most commonly affected of the large systemic arteries.11 Aortic tumors can be further classified by location as intraluminal, inti- mal, mural, or adventitial, with intimal being the most common (70%) followed by mural (20%).8,10,11 Intimal tumors of the aorta tend to spread along the intimal surface and frequently extend into the lumen to form a polypoid mass, as described in the present canine cases, whereas mural tumors tend to grow outward and invade adjacent structures.8,11 There are cases of aortic chondroid hyperplasia in other animal species, including rabbits; however, these lesions are limited to the intima of the aorta and rarely form intra-luminal projections or masses.2,12 Sarcomas of the aorta are likely derived from intimal cells that show myofibroblastic differentiation.7 Intimal cells have a variable capacity to differentiate to endothelial, myoid, or fibroblastic lineages, lending a more complex and variable phenotype to intimal neoplasms.11
An alternate consideration of the mass in the first case is of non-neoplastic chondroid hyperplasia or metaplasia. Histologically, the features are similar for these two processes, however, it is atypical for a hyperplastic chondroid-derived process to form a large polypoid and obstructive mass.2,12 Chondroid metaplasia can arise in the aortic media as a consequence of repeated trauma, previous inflammation, hypoxia, or chronic systemic disease such as conditions leading to atherosclerosis.13,14 It is possible that these present neoplasms arose out of metaplastic lesions; however, an original, local, or systemic vascular insult was not apparent in either of the discussed cases.
Clinical presentations in cases of aortic neoplasia in humans, similar to those in dogs, tend to include pain and decreased femoral pulses.9,11 Aortic neoplasms should be considered in cases of undetermined thromboembolic disease or unexplained weakness/lethargy. This disease should also appear in differential diagnoses for animals that present with hind limb paresis, pain, or with unilateral or bilateral absent pulses in the rear limbs. Obstructive intraluminal aortic masses should also be considered in dogs with cardiac failure and left ventricular systolic dysfunction, as in the first case. Echocardiography is a critical tool for diagnosis and care should be taken to carefully evaluate the aortic arch in order to avoid missing an intraluminal lesion. Cross-sectional imaging (CT, MRI) could be considered if clinical suspicion of an intravascular neoplasm is high, but was not pursued in these cases.
Conflicts of interest
The authors declare no conflicts of interest relative to this work.
Acknowledgments
The authors acknowledge Dr. Edward S. Cooper for assistance with medical care of the first case and Dr. John D. Bonagura for assistance with the medical care of the second case. The assistance of Dr. Doug Walther in obtaining the postmortem specimen from the second case is also gratefully acknowledged.
References
- Connolly HM, Oh JK, Schaff HV, Roger VL, Osborn SL, Hodge DO, Tajik AJ. Severe aortic stenosis with low transvalvular gradient and severe left ventricular dysfunction: result of aortic valve replacement in 52 patients. Circulation 2000;16:1940-1946.
- Anderson WI, Carberry CA, King JM, Trootter EJ, de Lahunta A. Primary aortic chondrosarcoma in a dog. Vet Pathol 1988;25:180-181.
- Callanan JJ, McCarthy GM, McAllister H. Primary pulmonary artery leiomyosarcoma in an adult dog. Vet Pathol 2000;37: 663-666.
- Lee B, Lee S, Lee H, Kim H, Kim D, Choi J. Abdominal aortic chondrosarcoma in a dog. J Vet Med Sci 2011;73: 1473-1476.
- Ranck RS, Linder KE, Haber MD, Meuten DJ. Primary inti- mal aortic angiosarcoma in a dog. Vet Pathol 2008;45: 361-364.
- Cohen JA, Bulmer BJ, Patton KM, Sisson DD. Aortic dissection associated with an obstructive aortic chondrosarcoma in a dog. J Vet Cardiol 2010;12:203-210.
- Burke AP, Virmani P. Sarcomas of the great vessels: a clin- icopathologic study. Cancer 1993;71:1761-1773.
- Busby JR, Ochsner JL, Emory B, Flaum M, Mitchell W. Malignant fibrous histiocytoma arising from descending thoracic aorta. Ann Vasc Surg 1990;4:185-188.
- Chen KTK. Primary malignant fibrous histiocytosis of the aorta. Cancer 1981;48:840-844.
- Restrepo CS, Betancourt SL, Martinez-Jimenez S, Gutierrez FR. Aortic tumors. Semin Ultrasound CT MR 2012; 3:265-272.
- Sebenik M, Ricci A, DiPasquale B, Mody K, Pytel P, Jee KL, Knuutila S, Scholes J. Undifferentiated intimal sarcoma of large systemic blood vessels. Am J Surg Pathol 2005;29: 1184-1193.
- Hadjiisky P, Donev S, Renais J, Scebat L. Cartilage and bone formation in arterial wall: morphological and histochemical studies. Basic Res Cardiol 1979;74:649-662.
- Chen M, Gatalica Z, Wang B. Cartilaginous and Osseous metaplasia in the aortic valve. Internet J Cardiol 2006;4: 1-4.
- Woldt E, Terrand J, Mlih M, Matz RL, Bruban V, Coudane F, Foppolo S, El Asmar Z, Chollet ME, Ninio E, Bednarczyk A, Thierse D, Schaeffer C, Van Dorsselaer A, Boudier C, Whahli W, Chambon P, Metzger D, Herz J, Boucher P. The nuclear hormone receptor PPARg counteracts vascular calcification by inhibiting Wnt5a signaling in vascular smooth muscle cells. Nat Commun 2012;3:1077.
^Наверх









