Analytical validation and clinical evaluation of a commercially available high-sensitivity immunoassay for the measurement of troponin I in humans for use in dogs

Author information
Winter R.L., Saunders A.B., Gordon S.G., Miller M.W., Sykes K.T., Suchodolski J.S., Steiner J.M. Analytical validation and clinical evaluation of a commercially available high-sensitivity immunoassay for the measurement of troponin I in humans for use in dogs // J Vet Cardiol. 2014 Jun;16(2):81-9.
Abstract
OBJECTIVE: To analytically validate a commercially available high-sensitivity immunoassay for measurement of cardiac troponin I (cTnI) in humans for use in dogs and to evaluate serum cTnI concentrations in healthy dogs and 3 well-defined groups of dogs with common cardiac diseases.
ANIMALS: Canine serum samples were used for validation. 85 client-owned dogs including 24 healthy controls, 20 with myxomatous mitral valve disease, 19 with congenital heart disease, and 22 with arrhythmias.
METHODS: Four serum samples were used to analytically validate the ADVIA Centaur TnI-Ultra assay by assessing intra-assay variability, inter-assay variability, spiking recovery, and dilutional parallelism. Dogs were grouped based on examination, echocard Analysis of the serum samples used for validation revealed an intra-assay coefficient of variation between 3.6% and 5.7%, and an inter-assay coefficient of variation between 2.4% and 5.9%. Observed to expected ratios for spiking recovery were 97.9 ± 8.6% (mean, SD). Observed to expected ratios for dilutional parallelism were 73.0 ± 11.5% (mean, SD). Dogs with cardiac disease had significantly higher serum cTnI concentrations (P < 0.005) than healthy dogs.
CONCLUSIONS: The ADVIA Centaur TnI-Ultra's low limit of detection allows measurement of serum cTnI in the majority of dogs even with no or mild cardiac disease. Dilution of samples for measurement of values above the upper limit of detection is not reliable and therefore not recommended. Serum cTnI concentrations are significantly higher in dogs with cardiac disease compared to healthy dogs.
Abbreviations
- ACVIM American College of Veterinary Internal Medicine cTnI cardiac troponin I
- CV coefficient of variation
- hs-cTnI high-sensitivity cardiac troponin I MMVD myxomatous mitral valve disease
Introduction
Cardiac troponin I (cTnI) is a cardiac-specific protein involved in cardiomyocyte contraction and relaxation.1 More specifically, it is one of 3 subunits of the cardiac troponin complex that plays a role in excitation—contraction coupling of the cardiomyocytes. Troponin I is also present in skeletal muscle, however, the cardiac-specific isoform of troponin I has a different amino acid sequence from its skeletal counterpart and both isoforms thus do not cross-immunoreact.1 Cardiac troponin I is normally bound to the actin filament and present in low concentration in the cytosol, but cardiomyocyte damage allows cTnI release into the extracellular space.1 Therefore, circulating concentrations of cTnI provide information about cardiac-specific injury. Canine and human cTnI share significant amino acid homology, thus cTnI assays designed for human patients can be used in canine patients with proper analytical validation.2 Circulating cTnI concentration has been used for the detection of both acute and chronic myocardial damage in multiple species, and is often utilized as adjunctive information to other diagnostic tests, such as thoracic radiography, electrocardiography, and echocardiography in case management.1—9
The concentration of circulating cTnI has been shown to be proportional to the degree of cardiomyocyte damage.1 Many different commercial assays are available, both standard sensitivity cTnI assays and high- sensitivity cTnI (hs-cTnI) assays. The main distinguishing characteristic of hs-cTnI assays is a lower level of cTnI detection compared to standard sensitivity cTnI assays which make them capable of detecting lower concentrations of circulating cTnI, potentially providing better sensitivity for detecting myocardial damage.10,11 In human medicine, cTnI assays can identify increases in serum cTnI in patients with acute diseases such as myocardial infarction,10,11 but also in chronic heart disease,12,13 congenital heart disease,14,15 and patients with clinically significant arrhythmias.16—18 In veterinary medicine, serum cTnI concentrations have been reported using standard sensitivity cTnI assays in dogs with myxomatous mitral valve disease (MMVD), dilated cardiomyopathy, arrhythmogenic right ventricular cardiomyopathy, and congenital heart disease.6,7,19—22 With standard cTnI assays, the lower limit of detection can be too high, limiting the ability to detect increases in cTnI and thereby limiting the assay’s clinical utility.7,21,23,24
The ADVIA Centaur TnI-Ultrad is a commercially available high-sensitivity 3-site assay for cTnI analysis that utilizes direct chemiluminometry with detectable cTnI serum concentrations reported by the manufacturer ranging from 0.006 to 50.0 ng/mL. This cTnI immunoassay has been previously validated for use in humans and cattle.25 The objectives of this study were to analytically validate the assay for use in dogs and to evaluate the clinical use of this assay in a population of healthy dogs and stratified in 3 well- defined groups of dogs with common cardiac diseases.
d ADVIA Centaur TnI-Ultra, Siemens Medical Solutions Diagnostics, New York, NY, USA.
Animals, materials and methods
Assay
Cardiac troponin I analysis was performed with the ADVIA Centaur TnI-Ultra immunoassay, a high- sensitivity 3-site assay using direct chem- iluminometry. One polyclonal goat antibody directed against antitroponin I and labeled with acridium ester and 2 biotinylated mouse monoclonal antibodies directed against antitroponin I are used for cTnI detection. These capture antibodies that recognize amino acid sequences 41—49 and 87—91 in the central region of the cTnI molecule.10,25 The solid phase reagent is made of magnetic latex particles that have been conjugated with strepta- vidin. The biotinylated antibodies bind the cTnI in the serum, and the biotin is then captured by the streptavidin-labeled magnetic particles. A direct relationship exists between the cTnI amount in the sample and the amount of relative light emitted from the chemiluminescent reaction that ensues.10,25 The working range for cTnI is reported by the manufacturer as 0.006—50.0 ng/mL. The amount of serum required for the assay is 100 mL. The manufacturer’s specifications for this assay allow the use of either plasma (heparinized or EDTA) or serum. The data reported herein are for serum samples.
Blood samples were collected from dogs presented to the Texas A&M University Veterinary Medical Teaching Hospital from October 2011 to December 2012. Blood was collected from the external jugular vein, and samples were placed into commercially available additive free glass tubes and allowed to rest for 15 min at room temperature to allow for complete blood clot formation. Serum was obtained via centrifugation and stored in 500—1000 mL aliquots in polypropylene tubes. Samples were immediately frozen and stored at -80 °C until analysis. Frozen samples were allowed to slowly thaw at room temperature immediately prior to batch analysis. Assay linearity was assessed using dilutional parallelism (i.e. assessing assay performance by diluting samples with subsequent cTnI measurement) of serial twofold dilutions from 1:2 to 1:8.26 Assay accuracy was evaluated by spiking different sera with other sera of known concentrations of cTnI. The percentages of observed to expected values for serial dilution and spiking recovery were calculated as [observed value (ng/mL)/expected value (ng/mL)] x 100.26 Precision was evaluated by the evaluation of selected serum samples 10 times within the same assay over a single day (i.e. within 1 assay plate) and calculating the intra assay coefficients of variation (CV).26 Reproducibility was determined by the evaluation of selected serum samples in 10 consecutive assay runs over 10 days (i.e. across multiple assay plates) and calculating inter-assay CV.26 Both the intraassay and inter-assay CV were calculated by % CV = [SD/mean] x 100.
Dogs
Serum from 10 client-owned dogs was submitted for cTnI analysis for the purpose of using some of these dogs’ samples for analytical assay validation. Ten mL blood samples were obtained from each dog, and the serum cTnI values were evaluated in these dogs. Samples from 4 of the dogs were chosen to be used for the analytical assay validation. These 4 samples were chosen specifically because they were found to have different serum cTnI concentrations that covered a range of cTnI values.
Serum samples were obtained from an additional 85 client-owned dogs that were presented for evaluation and assigned to 1 of 4 groups. Group 1 (G1-healthy) consisted of dogs that were considered apparently healthy with no abnormalities based on history, physical examination, echocardiogram, and laboratory analysis consisting of a complete blood-cell count and a serum biochemical analysis. The remaining 3 groups of dogs had cardiac disease and clinical evaluation consisted of a history, physical examination, and clinically indicated diagnostic tests. Dogs assigned to group 2 (G2-MMVD) had MMVD.27 Dogs assigned to group 3 (G3-congenital) had congenital heart disease. Dogs assigned to group 4 (G4-arrhythmia) had clinically relevant arrhythmias based on the presence of clinical signs and abnormalities on electrocardiography and 24- h ambulatory electrocardiography. The dogs in G4-arrhythmia had clinical signs directly caused by the arrhythmia.
This study was reviewed and approved by the Institutional Animal Care and Use Committee at Texas A&M University, College Station, TX.
Statistical analysis
The D’Agostino and Pearson omnibus test was used to test each data set for age, weight, and troponin concentration for normality using a commercially available software package.e Data sets that failed normality testing were expressed as medians and ranges, otherwise data were reported as mean ± SD. A Kruskal—Wallis one-way analysis of variance test was used to assess all data sets that failed normality testing for differences between groups. A Dunn’s multiple comparison test was used to test for differences between each group. A P-value < 0.05 was considered significant for all statistical analyses. Serum samples with cTnl concentrations below the lower limit of detection were assigned a value of 0.006 ng/mL.
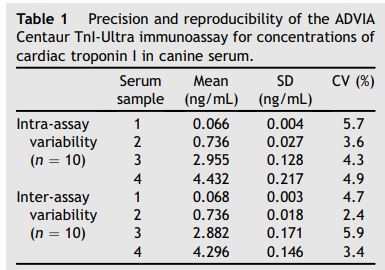
Table 1 Precision and reproducibility of the ADVIA Centaur TnI-Ultra immunoassay for concentrations of cardiac troponin I in canine serum.
e GraphPad Prism® (v.6.0), GraphPad Software, San Diego, CA, USA.
Results
Samples used for analytical assay validation had measured cTnl concentrations of 0.066, 0.768, 2.826, and 4.117 ng/mL. The 4 serum samples had intra-assay CV that ranged between 3.6 and 5.7% and inter-assay CV that ranged between 2.4 and 5.9% (Table 1).
Observed to expected ratios for serial dilutions for the 4 samples were 72.7 ± 11.5% (mean, SD) with a range of 60.4—94.3% at dilutions of 1 in 2, 1 in 4, and 1 in 8 (Table 2). Observed to expected ratios for spiking recovery were 97.9 ± 8.6% with a range of 87.0—109.6% for spiking each of the 4 serum samples with each of the other 3 serum samples (Table 3).
The samples from 85 client-owned dogs were evaluated and group characteristics are reported in Table 4. Serum cTnl concentrations were detectable in 79/85 dogs. Values were below the lower detection limit in 5 dogs from G1-healthy and 1 dog from G2-MMVD. Breeds in G1-healthy consisted of mixed (n = 4), labrador retriever (n = 3), border collie (n = 2), catahoula hog dog (n = 2), bull mastiff (n = 2), and all other breeds were represented by 1 dog each. Breeds in G2- MMVD consisted of Cavalier King Charles Spaniel (n = 14), and 1 each of mixed, border collie, dalmatian, Australian shepherd, and miniature schnauzer. Stage based on the American College of Veterinary Internal Medicine (ACVIM) guidelines included B1 (n = 7), B2 (n = 7), and C (n = 5). Dogs with MMVD ACVIM Stage C were considered clinically stable based on the absence of active pulmonary edema. One dog in G2-MMVD with the highest cTnI value of 5.393 ng/mL was diagnosed as MMVD ACVIM Stage C with mild pulmonary hypertension and multiform ventricular tachycardia that required anti-arrhythmic therapy. Based on the presence of a clinically important arrhythmia of unknown etiology complicating the diagnosis of MMVD ACVIM Stage C, this dog was not included in the statistical analysis. Breeds in G3- congenital consisted of boxer (n = 3), maltese (n = 2), Rhodesian ridgeback (n = 2), and all other breeds were represented by 1 dog each. Diagnoses of dogs in G3-congenital included moderate to severe pulmonic stenosis (n = 10), severe subaortic stenosis (n = 3), left-to-right shunting patent ductus arteriosus (n = 3), tricuspid valve dysplasia with concurrent atrial fibrillation (n = 2), and a restrictive left-to-right shunting ventricular septal defect (n = 1). Breeds in G4-arrhythmia consisted of boxer (n = 5), labrador retriever (n = 5), Doberman pinscher (n = 3), collie (n = 2), and all other breeds were represented by 1 dog each. Dogs in G4-arrhythmia had ventricular tachyarrhythmias (n = 15), supraventricular tachycardia (n = 5), and sick sinus syndrome (n = 1).
Table 2 Results for dilutional parallelism of the ADVIA Centaur TnI-Ultra immunoassay for measurement of cardiac troponin I in canine serum
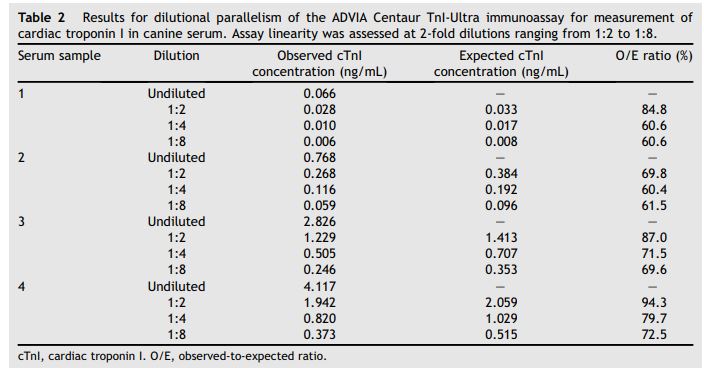
Assay linearity was assessed at 2-fold dilutions ranging from 1:2 to 1:8.
Table 3 Results of spiking recovery for the ADVIA Centaur TnI-Ultra immunoassay for canine serum was spiked into each of the other samples
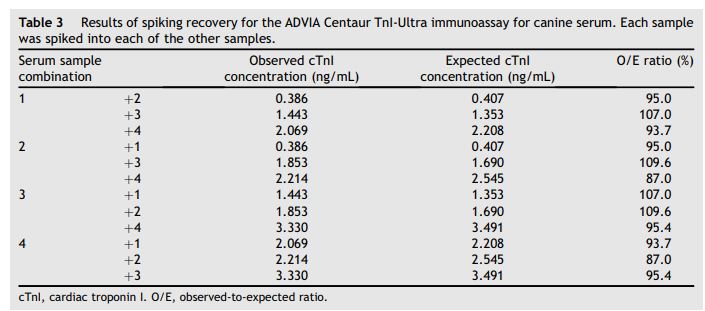
Each sample.
Dogs in G1-healthy and G3-congenital were both significantly (P < 0.05) younger than those in both G2-MMVD and G4-arrhythmia. There was no significant difference in age between dogs in G1- healthy and G3-congenital, or between dogs in G2-MMVD and G4-arrhythmia. Dogs in G2-MMVD weighed significantly (P < 0.05) less than those of G1-healthy and G4-arrhythmia, and those of G3- congenital weighed significantly less than those of G4-arrhythmia. There was no significant difference in weight between dogs of G2-MMVD and those of G3-congenital, between those of G1-healthy and those of both G3-congenital and G4-arrhythmia. Dogs with cardiac disease (G2-MMVD, G3- congenital, G4-arrhythmia) had significantly higher serum cTnI concentrations (P < 0.005) than healthy dogs (G1-healthy) (Fig. 1). There was no significant difference in serum cTnI concentrations between dogs of any of the 3 groups with cardiac disease (G2-MMVD, G3-congenital, or G4- arrhythmia).
Discussion
Our results indicate that the ADVIA Centaur TnI- Ultra immunoassay , a commercially available hs-cTnI assay for use in humans, can be used to accurately and reliably measure serum cTnI in dogs at lower detection limits. The majority of dogs with and without cardiac disease had measurable concentrations of cTnI using this assay. Five healthy dogs in G1-healthy and 1 dog with cardiac disease in G2-MMVD (MMVD - ACVIM Stage B1) had cTnI measurements below the lower limit of detection of this assay. Dogs with cardiac disease had significantly higher concentrations of cTnI than dogs without cardiac disease. Validation for this assay consisted of using 4 canine serum samples with different concentrations of cTnI to assess linearity, spiking recovery, intra-assay CV, and inter-assay CV. Intra-assay CV and inter-assay CV were each <10%, which is a precision classified as acceptable for clinical use by the International Federation of Clinical Chemistry and Laboratory Medicine.28,29 Based on the limited linearity observed, we do not recommend diluting samples for clinical analysis. However, data from this study would suggest that the range of 0.006-50.0 ng/mL is adequate for clinical assessment. Observed-to-expected ratios for serial dilutions ranged from 60.4 to 94.3%, and observed to expected ratios for spiking recovery ranged from 87.0 to 109.6%. These data indicate that although dilution occurred reliably, there was limited linearity of this assay when analyzing dog serum.
Table 4 Sex, age, weight, and serum cardiac troponin I concentrations (cTnI) for 84 client-owned dogs classified as healthy (G1-healthy) or having myxomatous mitral valve disease (G2-MMVD), congenital heart disease (G3- congenital), or clinically relevant arrhythmias (G4-arrhythmia)
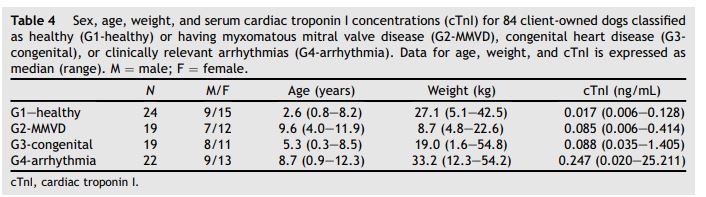
Data for age, weight, and cTnI is expressed as median (range). M = male; F = female.
Figure 1 Serum cardiac troponin I (cTnl) concentrations in healthy dogs (G1), dogs with myxomatous mitral valve disease (G2), dogs with congenital cardiac disease (G3), and dogs with clinically relevant arrhythmias (G4)
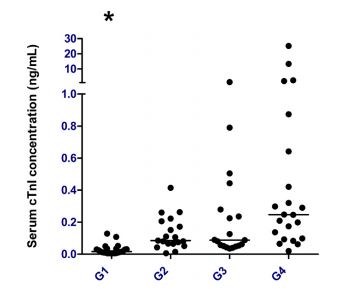
There was no significant difference in serum cTnI concentrations between dogs in any of the 3 groups with cardiac disease (G2, G3, or G4). Dogs with cardiac disease had significantly higher serum cTnI concentrations than healthy dogs (G1). Lines depict the median for each group. P values < 0.05 were considered significant. *denotes a significant difference compared to the 3 other groups.
The sample diluent used was provided by the manufacturer of the assay. This diluent is designed for use with human serum. Although human and canine cTnI have 94.3% amino acid homology,2 it is unknown how this diluent manufactured to be used with human serum interacts with canine cTnI. Additionally, sources of endogenous interference in the assay’s performance such as sample hemolysis, lipemia, or serum proteins have been shown to affect data interpretation in human studies,30,31 and it is unknown whether similar endogenous interferences were present in any of the canine samples in this study. Further investigation into these areas is warranted.
In a pilot study by the authors, both EDTA and heparinized plasma showed consistently lower concentrations of cTnI than in serum in dogs (data not shown). A previous study has shown that the addition of heparin to human serum cTnI samples decreases the ability of some assays to detect cTnI, and it is possible that this also occurs for canine plasma with the ADVIA Centaur TnI-Ultra immunoassay.30 Further investigation into this difference is warranted. This may prove important clinically in the context of serial measurements for assessment of disease progression, or regarding samples collected at multiple hospitals for one individual animal. Additionally, a previous study has demonstrated cardiac troponin T stability at -70 °C for at least one year.32
Serum cTnI concentrations in groups 2-4 (i.e. dogs with cardiac disease) were significantly higher when compared to the healthy control dogs in G1-healthy. This is a similar finding of previous studies evaluating cTnI between groups of dogs using other assays.7,33-35 However, previous veterinary studies have used assays that failed to detect measurable concentrations of cTnI in normal dogs, thus complicating direct comparison between dogs with and without cardiac dis- ease.7,21,23,33 The data reported in this study may provide a more reliable comparison of cTnI in healthy and dogs with cardiac disease, as roughly 80% (N = 19/24) of healthy dogs had serum cTnI concentrations at a measurable concentration using the ADVIA Centaur TnI-Ultra immunoassay. Additionally, cardiac diseases with mild car- diomyocyte damage such as MMVD may benefit from evaluation with this immunoassay. Two veterinary studies in which this cTnI immunoassay was used reported similar values for normal dogs (mean 0.020 ng/mL36 and mean 0.015 ng/mL24) compared to the data reported in this study. A recent publication demonstrated the clinical utility of the ADVIA Centaur Ultra-TnI assay in normal and clinically-affected dogs and cats using a validation process different from the process reported in this study.36 This previous study36 only used 2 dog breeds (labrador retrievers and Newfoundlands) from which to measure cTnI for a healthy dog reference range. The study reported herein utilized a wide spectrum of dog breeds, ages, and sizes applicable to a greater population of dogs and useful for widespread reference. The previous paper36 compares clinically abnormal dogs to healthy dogs without providing details regarding signalment and disease severity. The study reported herein also benefits from utilizing 3 groups of dogs with different and commonly-encountered cardiac disease states. Additionally, dogs in this study with MMVD were further categorized into the ACVIM staging system. Previous studies indicate that dogs that are older have higher serum cTnI concentrations than younger dogs with similar disease processes.6,8,34
However, this is unlikely to account for the greater cTnI in dogs with cardiac disease in this study considering dogs with congenital heart disease in G3-congenital were significantly younger than dogs with MMVD and arrhythmias in G2-MMVD and G4-arrhythmia, respectively. Additionally, there was no significant difference in age between dogs in G1-healthy and G3-congenital. It is also unlikely that the weight of the dogs impacted differences in serum cTnI concentrations between groups, considering dogs in G2-MMVD weighed significantly less than dogs in G1-healthy. Dogs and humans with renal failure have significantly higher cTnI concentrations compared to healthy subjects.37-39 However, the circulating serum cTnI concentration does not directly correlate with the degree of azotemia.37,39 The exact mechanism through which renal function affects circulating serum cTnI concentrations is unclear.
A limitation of this study includes the number of dogs represented from each specific disease. Although the 4 groups of dogs in this study were classified based on underlying cardiac disease category there were insufficient numbers of dogs in each individual group to allow for the evaluation of subsets of dogs within the groups. Forexample there may be important differences between dogs with different forms of congenital heart disease, and type of arrhythmia. Previous studies have reported a correlation with cTnI and stage of MMVD.6,8 However, it was not the goal of this study to generate reference ranges for specific forms of canine cardiac disease. Nor was it the goal of this study to assess the influence of renal and hepatic disease. An additional limitation is the lack of assessment of the correlation of histologic analysis and post-mortem examination to cTnI concentrations that has been previously described for dogs with MMVD.40 Lastly, although dogs were considered healthy based on no historical evidence of clinical disease, no significant abnormalities on their CBC or biochemical analysis, and no cardiac abnormalities detected during auscultation or echocardiography, it is possible that some of the dogs classified as healthy had underlying cardiac or systemic disease not identified at the time of cTnI sampling. Some dogs in our study classified as healthy without cardiac abnormalities may have sustained mild cardiomyocyte damage based on cTnI values and previous veterinary studies in which this cTnI immunoassay was used.24,36 Variations in study methods and signalment characteristics makes comparisons between these studies difficult.
Conclusions
In conclusion, the ADVIA Centaur TnI-Ultra immunoassay intended for use in humans performs adequately for measuring serum cTnI concentrations in dogs with and without cardiac disease. Based on the limited linearity of this assay, diluting values above the upper limit of the working range (i.e. over 50 ng/mL) is not recommended. Dogs with cardiac disease have significantly higher serum cTnI concentrations than healthy dogs based on the dogs studied here. We conclude that this immunoassay is sufficiently accurate, precise, and reproducible for clinical use in dogs. Moreover, the results of this immunoassay validation should allow clinicians to use the ADVIA Centaur TnI-Ultra immunoassay without concern for erroneous cTnI concentrations in dogs. The results of this study, and the lower limit of detection combined with high level of precision of this assay, suggests that most dogs with or without cardiac disease will have cTnI concentrations with reportable values when measured using this assay.
Conflict of interest statement
The authors have no conflict of interest.
References
- O'Brien PJ, Smith DEC, Knechtel TJ, Marchak MA, Pruim- boom-Brees I, Brees DJ, Spratt DP, Archer FJ, Butler P, Potter AN, Provost JP, Richard J, Snyder PA, Reagan WJ. Cardiac troponin is a sensitive, specific biomarker of cardiac injury in laboratory animals. Lab Anim 2006;40: 153-171.
- Rishniw M, Barr SC, Simpson KW, Winand NJ, Wootton JA. Cloning and sequencing of the canine and feline cardiac troponin I genes. Am J Vet Res 2004;65:53-58.
- Alpert JS, Tyygesen K, Jaffe A, White HD. Universal definition of myocardial infarction: a consensus document: ischaemic heart disease. Heart 2008;94:1335-1341.
- Nellessen U, Goder S, Schobre R, Abawi M, Hecker H, Tschoke S. Serial analysis of troponin I levels in patients with ischemic and nonischemic dilated cardiomyopathy. Clin Cardiol 2006;29:219-224.
- Tanindi A, Cemri M. Troponin elevation in conditions other than acute coronary syndrome. Vasc Health Risk Manag 2011;7:597-603.
- Oyama MA, Sisson DD. Cardiac troponin I concentration in dogs with cardiac disease. J Vet Intern Med 2004;18: 831-839.
- Wess G, Simak J, Mahling M, Hartmann K. Cardiac troponin I in Doberman pinschers with cardiomyopathy. J Vet Intern Med 2010;24:843-849.
- Ljungvall I, Hoglund K, Tidholm A, Olsen LH, Borgarelli M, Venge P, Haggstrom J. Cardiac troponin I is associated with severity of myxomatous mitral valve disease, age, and C-reactive protein in dogs. J Vet Intern Med 2010;24: 153-159.
- Suzuki K, Uchida E, Schober KE, Niehaus A, Rings MD, Lakritz J. Cardiac troponin I in calves with congenital heart disease. J Vet Intern Med 2012;26:1056-1060.
- Casals G, Filella X, Bedinin JL. Evaluation of a new ultrasensitive assay for cardiac troponin I. Clin Biochem 2007;40: 1406-1413.
- Clerico A, Giannoni A, Prontera C, Giovanni S. High-sensitivity troponin: a new tool for pathophysiological investigation and clinical practice. Adv Clin Chem 2009;49: 1 -30.
- Nagarajan V, Hernandez AV, Tang WHW. Prognostic value of cardiac troponin in chronic stable heart failure: a systematic review. Heart 2012;98:1778-1786.
- Omland T, Pfeffer MA, Solomon SD, de Lomos JA, Rosjo H, Saltyte BJ, Maggioni A, Domanski MJ, Rouleau JL, Sabatine MS, Braunwald E, PEACE Investigators. Prognostic value of cardiac troponin I measured with a highly sensitive assay in patients with stable coronary artery disease. J Am Coll Cardiol 2013;61:1240-1249.
- Sugimoto M, Ota K, Kajihama A, Nakau K, Manabe H, Kajino H. Volume overload and pressure overload due to left-to-right shunt-induced myocardial injury: evaluation using a highly sensitive cardiac troponin I assay in children with congenital heart disease. Circ J 2011 ;75: 2213-2219.
- Correale M, Nunno L, Ieva R, Rinaldi M, Maffei G, Magaldi R, Di Biase M. Troponin in newborns and pediatric patients. Cardiovasc Hematol Agents Med Chem 2009;7: 270-278.
- Conti A, Mariannini Y, Viviani G, Poggioni C, Cerini G, Luzzi M, Zanobetti M, Innocenti F, Padeletti L, Gensini GF. Abnormal troponin level as short-term predictor of poor outcome in acute atrial fibrillation. Am J Emerg Med 2013; 31:699-704.
- Chow GV, Hirsch GA, Spragg DD, Cai JX, Cheng A, Ziegelstein RC, Marine JE. Prognostic significance of cardiac troponin I levels in hospitalized patients presenting with supraventricular tachycardia. Medicine (Baltimore) 2010; 89:141-148.
- Gupta S, Pressman GS, Figueredo VM. Incidence of, predictors for, and mortality associated with malignant ventricular arrhythmias in non-ST elevation myocardial infarction patients. Coron Artery Dis 2010;21:460-465.
- Baumwart RD, Orvalho J, Meurs KM. Evaluation of serum cardiac troponin I concentration in Boxers with arrhyth- mogenic right ventricular cardiomyopathy. Am J Vet Res 2007;68:524-528.
- Saunders AB, Smith BE, Fosgate GT, Suchodolski JS, Steiner JM. Cardiac troponin I and C-reactive protein concentrations in dogs with severe pulmonic stenosis before and after balloon valvuloplasty. J Vet Cardiol 2009; 11:9-16.
- Guglielmini C, Civitella C, Diana A, Di Tommaso M, Cipone M, Luciani A. Serum cardiac troponin I concentration in dogs with precapillary and postcapillary pulmonary hypertension. J Vet Intern Med 2010;24:145-152.
- Hezzell MJ, Boswood A, Chang YM, Moonarmart W, Souttar K, Elliott J. The combined prognostic potential of serum high-sensitivity cardiac troponin I and N-terminal pro-B-type natriuretic peptide concentrations in dogs with degenerative mitral valve disease. J Vet Intern Med 2012; 26:302-311.
- Verbiest T, Binst D, Waelbers T, Coppieters E, Polis I. Perioperative changes in cardiac troponin I concentrations in dogs. Res Vet Sci 2013;94:446-448.
- Serra M, Papkonstantinou S, Adamcova M, O'Brien PJ. Veterinary and toxological applications for the detection of cardiac injury using cardiac troponin. Vet J 2010;185: 50-57.
- Varga A, Schober KE, Walker WL, Lakritz J, Rings MD. Validation of a commercially available immunoassay for the measurement of bovine cardiac troponin I. J Vet Intern Med 2009;23:359-365.
- Steiner JM, Williams DA. Development and analytical validation of an enzyme linked immunosorbent assay for the measurement of canine gastric lipase immunoreactivity in serum. Can J Vet Res 2004;68:161-168.
- Atkins C, Bonagura J, Ettinger S, Fox P, Gordon S, Haggstrom J, Keene B, Luis-Fuentes V, Stepien R. Guidelines for the diagnosis and treatment of canine chronic valvular heart disease. J Vet Intern Med 2009;23:1142-1150.
- Tate JR. Troponin revisited 2008: assay performance. Clin Chem Lab Med 2008;46:1489-1500.
- Pateghini M, Gerhardt W, Apple FS, Dati F, Ravkilde J, Wu AH. Quality specifications for cardiac troponin assays. Clin Chem 2001;39:175-179.
- Katrukha A, Bereznikova A, Filatov V, Esakova T. Biochemical factors influencing measurement of cardiac troponin I in serum. Clin Chem Lab Med 1999;37:1091-1095.
- Dimeski G. Interference testing. Clin Biochem Rev 2008;29: S43-S48.
- Basit M, Bakshi N, Hashem M, Allebban Z, Lawson N, Rosman HS, Maciejko JJ. The effect of freezing and longterm storage on the stability of cardiac troponin T. Am J Clin Pathol 2007;128:164-167.
- Spratt DP, Mellanby RJ, Drury N, Archer J. Cardiac troponin I: evaluation of a biomarker for the diagnosis of heart disease in the dog. J Small Anim Pract 2005;46:139-145.
- Oyama MA, Solter PF. Validation of an immunoassay for the measurement of canine cardiac troponin I. J Vet Cardiol 2004;6:17-24.
- Silvestrini P, Piviani M, Alberola J, Rodriguez-Cortes A, Planellas M, Roura X, O'Brien PJ, Pastor J. Serum cardiac troponin I concentrations in dogs with leishmaniasis: correlation with age and clinicopathologic abnormalities. Vet Clin Pathol 2012;41:568-574.
- Langhorn R, Willesen JL, Tarnow I, Kjelgaard-Hansen M. Evaluation of a high-sensitivity assay for measurement of canine and feline serum cardiac troponin I. Vet Clin Pathol 2013 [Epub ahead of print].
- Porciello F, Rishniw M, Herndon WE, Birettoni F, Antognoni MT, Simpson KW. Cardiac troponin I is elevated in dogs and cats with azotemia, renal failure, and in dogs with non-cardiac systemic disease. Aust Vet J 2008;86:390-394.
- Freda BJ, Tang WHW, Van Lente F, Peacock WF, Francis GS. Cardiac troponins in renal insufficiency. J Am Coll Cardiol 2002;40:2065-2071.
- Sharkey LC, Berzina I, Ferasin L, Tobias AH, Lulich JP, Hegstad-Davies RL. Evaluation of serum cardiac troponin I concentration in dogs with renal failure. J Am Vet Med Assoc 2009;234:767-770.
- Falk T, Ljungvall I, Zois NE, Hoglund K, Olsen LH, Pederson HD, Haggstrom J. Cardiac troponin I concentration, myocardial arteriosclerosis, and fibrosis in dogs with congestive heart failure because of myxomatous mitral valve disease. J Vet Intern Med 2013;27: 500-506.
^Наверх









