The evolution of the natriuretic peptides - Current applications in human and animal medicine

Author information
van Kimmenade RR1, Januzzi JL Jr.The evolution of the natriuretic peptides - Current applications in human and animal medicine. J Vet Cardiol. 2009 May;11 Suppl 1:S9-21.
Abstract
Although natriuretic peptides have played an important role in the fluid homeostasis of vertebrates for over several million years, their importance has only been noticed in the last few decades. Yet, the family of natriuretic peptides have since their discovery, drawn the attention of a broad spectrum of physicians and researchers involved in the maintenance of fluid homeostasis, including marine biologists, basic scientists, physicians and veterinarians. While all natriuretic peptides share a common phylogenetic background, due to differences in receptor-binding affinities, they have evolved into different hormones with clear distinct functions. B-type natriuretic peptide (BNP) is the most studied member of the natriuretic peptide family , and together with its cleavage equivalent amino-terminal proB-type natriuretic peptide (NT-proBNP) these peptides have emerged as important cardiovascular serum markers. However, since their introduction, physicians involved in human or animal medicine have faced common but also different challenges in order to optimally interpret the diagnostic and prognostic value of these novel cardiovascular biomarkers.
Introduction
In 1981, de Bold et al. reported their fascinating discovery that the injection of atrial tissue extracts peptide (ANP)2 stimulated a flood of research leading to the discovery of a complete new class of hormones, the natriuretic peptides (NP).
The family of natriuretic peptides is a group of phylogenetically, functionally and structurally related peptides in vertebrates, whose main function is the regulation of fluid homeostasis.3 The known members of the NP family thus far are ANP with its renal alter-ego urodilatin, B-type natriuretic peptide (BNP), C-type natriuretic peptide in 4 different isoforms, dendroaspis natriuretic peptide (DNP) and ventricular natriuretic peptide (VNP), the latter probably only expressed in fish. The structures of these peptides are depicted in Fig. 1. All NPs are synthesized as pre-pro-hormones, and have a 17- amino acid ring structure with a common amino- terminal tail; with the exception of CNP isotypes, all natriuretic peptides have a carboxy-terminal portion as well.4
ANP, BNP and VNP are primarily produced by cardiomyocytes, CNP is mostly expressed in the brain and endothelium, whilst dendroaspis natriuretic peptide is up till now only found in the venom of the Green Mamba snake.4 The actions of the NPs are mediated by a set of specific natriuretic peptide receptors (NPR), and variations in the effects of the different NPs primarily depend on the differences in local expression and production. However their degra¬dation and clearance rates are important as well. The utility of the natriuretic peptides in clinical practice is now intensively studied, not only as diagnostic and prognostic serological biomarkers in cardiovascular diseases, but also as novel therapeutic agents; although somewhat lagging behind the under¬standing for human applications, use of natriuretic peptides, B-type natriuretic peptide and especially its amino-terminal pro-hormone, NT-proBNP is now more widespread in veterinary medicine.
The ABC of natriuretic peptides
ANP is primarily produced in both atria: production and release of ANP is typically triggered by increased atrial-wall tension following an increase of intravascular volume or pressure. In contrast to BNP, whose production is rapidly up-regulated when cardiomyocytes are stimulated, ANP is stored in granules which release their content into the circulation after stimulation.5 The ventricles of the normal adult heart produce only small amounts of ANP, but this production is remarkably enhanced in the ventricular tissue of fetuses/ neonates, in hypertrophied ventricles and in situ¬ations of volume overload.1,3,6
Besides production in the atria, another version of ANP is produced in the kidneys called urodilatin. This peptide was originally isolated in urine, and due to an alternative splicing of the predecessor molecule proANP, it bears a 4-amino acid longer amino-terminal tail, when compared to its atrial counterpart.7 Urodilatin is produced in the renal distal tubules and only has a local function in the sodium and volume regulation in the kidneys. Like all cardiac NPs, urodilatin decreases the renin secretion from the macula densa and inhibits aldosterone release from the zona glomerulosa and the angiotensin II-stimulated proximal tubular sodium and water reabsorption.8
Figure 1 The different members of the natriuretic peptide family. ANP = atrial natriuretic peptide; BNP = B-type natriuretic peptide; CNP = C-type natriuretic peptide; DNP = dendroaspis natriuretic peptide; VNP = ventricular natriuretic peptide.

BNP, now called B-type natriuretic peptide (but originally identified in the porcine brain and called brain natriuretic peptide), is found wherever myocardial tissue is present but particularly in the ventricles.9,10 In contrast to ANP which has (in humans) a half-life in vivo of 3 min, due to a lower sensitivity for both the clearance receptor and degrading enzymes, B-type natriuretic peptide is a more stable peptide with a half-life in vivo in humans of 20 min providing greater ease in its measurement.11,12 BNP is rapidly produced by cardiomyocytes after stimuli like myocardial stretch or hypoxia.13,14 Most of the effects of BNP are also mediated by the other cardiac natriuretic peptides but since B-type natriuretic peptide is the more stable counterpart in humans, it is by far the most documented family member.
After its release, B-type natriuretic peptide induces diuresis and natriuresis via direct tubular actions as described above and via an increase of the glomerular filtration,3 while it may also inhibit the cardiac sympathic nervous system and renine—angio¬tensin—aldosterone system by suppressing norepi¬nephrine and aldosterone levels.3,15,16 In addition, BNP has shown to exert anti-proliferative, anti- fibrotic as well as relaxing actions on heart and vascular tissue, providing a cardiovascular protection mechanism.17—19 Although its production is up-regulated in situ¬ations of volume overload, the diuretic and natriuretic properties of BNP in (patho)physiological concentrations are still insufficient to retain euvolemia. Yet, since serum concentrations are elevated in hypervolemia, B-type natriuretic peptide and its cleavage equivalent NT-proBNP have shown to be powerful biomarkers for the diagnosis and prognosis of cardiovascular diseases.20
CNP is a paracrine or autocrine factor and predominates in the vascular endothelial cells, the central nervous system, the kidney, chondrocytes and the pituitary gland of mammals. Plasma concentrations are low but tissue expression is high, consistent with a paracrine role.21,22 CNP is considered to have a neurotransmitter function in the central nervous system while it also plays a role in the regulation of the vascular tone.23,24 Furthermore, CNP inhibits fibroblast activation and prevents the development of fibrosis in a manner superior to ANP or BNP, but it lacks significant natriuretic or diuretic actions.21,25 Besides, CNP also plays an important role in endochondral ossification, testicular endocrine function, sper¬matogenesis and adequate placental function during pregnancy.26—28
Dendroaspis natriuretic peptide is a unique NP, which thus far has only been isolated from the venom gland of the Dendroaspis angusticeps, the Green Mamba.29 DNP is structurally very similar to VNP in terms of long C-terminal 'tail’ sequence (see Fig. 1). Although its function thus far remains unknown, the vasorelaxing effect of DNP may potentiate the uptake of the neuro¬toxic venom of the Green Mamba.
Interestingly, specific anti-sera raised against DNP resulted in the measurement of DNP-like immunoreactivity in the human heart, while it has been demonstrated that circulating levels of dendroaspis natriuretic peptide - like peptides corresponded to the severity of heart failure in humans; therapeutic applications of DNP are now studied as well.30—32 Further research will be necessary in order to elucidate the exact role of DNP in vertebrates. VNP plays an important role in the fluid homeostasis of primitive ray-finned bony fish such as eels and salmonids and is abundantly expressed in these hearts. This is because fluid and salt homeostasis in these species are more challenging than in most terrestrial animals due to the changes in environment during their life span, and VNP usually coexists in combination with B-type natriuretic peptide, ANP and/ or CNP.4 Compared to other NPs, VNP has a long 14-amino acid carboxy-terminal tail and a high affinity for both the NPR-A and NPR-C.
The family history of the natriuretic peptides
Vertebrates are considered to have evolved from invertebrate chordates in salt water, who subse¬quently entered fresh water prior to returning to the seas or moving onto land.33 During the transi¬tion from aquatic to terrestrial life, sea animals encountered desiccation and gravity; it is thought that in response such animals developed mecha¬nisms to conserve body water and acquired powerful hearts and high blood pressure to main tain circulation in the face of gravity.4
To better grasp the phylogenetic background of the NPs it is necessary to understand that the genome is thought to have duplicated three times during vertebrate evolution: the first time (T1) at the transition from chordates to vertebrates, the second time (T2) at the transition from jawless fish (cyclostomes) to jawed vertebrates (gnathos- tomes), and the third time (T3) after divergence of the ray-finned As mentioned previously, 4 isoforms of CNP have been isolated in vertebrates. The isoform now known as CNP4, is believed to be the ancestral member of the NP family. CNP4 duplicated at T2 in order to generate the CNP3 gene and is silenced afterwards again in cartilaginous fish. It is not known whether the heart-specific NP genes (ANP, BNP and VNP) already existed in cartilaginous fish, but the abundant expression of the CNP3 gene in their hearts indicates that CNP3 acted as a cardiac NP before the CNP3 gene evolved into the ANP, B-type natriuretic peptide and VNP genes.4 CNP2 and CNP1 were only identi¬fied in ray-finned bony fish (with the exception of CNP1 expression that was found in chickens as well) and these genes are probably duplicated from the CNP3 gene at T3.34,35
Figure 2 The evolutionary history of the natriuretic peptides in vertebrates.
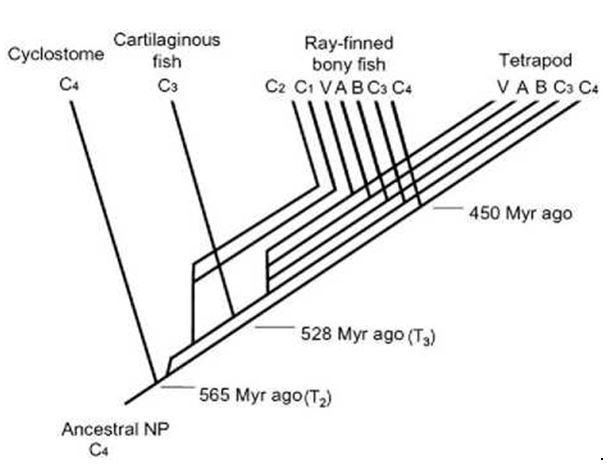
A = ANP, B = BNP, Cx = CNPx isoform, V = VNP, Myr = million years, NP = natriuretic peptide; Tx = time x. Adapted from Inoue et al.35 with permission.
ANP, BNP and VNP originate from CNP3. These genes are expressed almost exclusively in the heart and are released into the circulation in order to regulate body fluid homeostasis.3 Their relative importance may be increased in tetrapods, in which the heart maintains a higher performance to keep blood circulating against gravity. It seems that several NP genes have disappeared in tetra- pods, particularly CNP genes, while cardiac NP genes are retained. The VNP gene is only detected in primitive ray-finned fish species such as the eel and salmonids, and parallel to the presence of ANP and BNP, suggesting that the VNP gene is lost during teleost evolution.36 Although eels and salmonids may not need a strong cardiac function in order to pump blood against gravity, their habitats during their life span alter between fresh and salt water and the cardiac NPs in these fish contribute to body fluid regulation. Interestingly, since ANP was undetectable in the heart and the genome of Japanese medaka (a ray-finned bony fish species), BNP is the only cardiac NP common to all teleost species. In tetrapods, all mammalian and amphibian species thus far examined express both ANP and B-type natriuretic peptide in their hearts, yet birds and crocodiles lack the expression of ANP. Therefore, since all vertebrate species express BNP, it is assumed that BNP (and not ANP as previously postulated), is the basic cardiac NP in vertebrates.
Natriuretic peptide receptors and neutral endopeptidase
Four subtypes of NPRs have been identified, namely NPR-A, NPR-B, NPR-C and NPR-D (see Fig. 3). All NPRs have an extracellular ligand¬binding domain and a single transmembrane region in common.
Figure 3 The natriuretic peptide receptors.
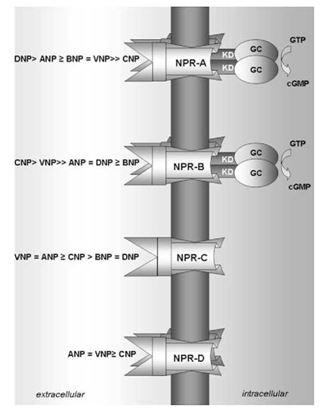
NPR- A = natriuretic peptide receptor type A; NPR-B = na¬triuretic peptide receptor type B; NPR-C = natriuretic peptide receptor type C; NPR-D = natriuretic peptide receptor type D; GTP = guanosine triphosphate; cGMP = cyclic guanosine monophosphate; KD = kinase homology domain; GC = guanylyl cyclase domain; ANP = atrial natriuretic peptide; BNP = B-type natri¬uretic peptide; CNP = C-type natriuretic peptide; DNP = dendroaspis natriuretic peptide; VNP = ven¬tricular natriuretic peptide.
However, there are some distinctions between the receptors: NPR-A and NPR-B contain an intracellular kinase homology and guanylyl cyclase domain, while NPR-C and NPR-D lack these intracellular domains. Furthermore, NPR-A, NPR-B and NPR-D are expressed as tetrameres while NPR- C is expressed as adimere.37 Specific characteris¬tics of the different receptors are discussed below.
NPR-A
The NPR-A is expressed in the lungs, brain, heart, adrenal glands, kidneys and the terminal ileum.38 The stimulation of the ligand-binding domain of NPR-A leads to the production of the intracellular second messenger cGMP, as necessary product for further signal transduction. NPR-A stimulation finally results in natriuresis, inhibition of renin and aldosterone, as well as vasorelaxant, anti-fibrotic, anti-hypertrophic and lusitropic effects.39 NPR-A knock-out animals develop high blood pressure, left ventricular hypertrophy and fibrosis.38,40 The sensitivity of the NPR-A for the different NPs ranks from high to low sensitivity as follows: DNP > ANP > BNP = VNP >> CNP.30,41—43
NPR-B
The NPR-B is expressed in the lungs, brain, skin, adrenal glands, kidneys, uterus and ovaries. The structure of NPR-B is similar to NPR-A, and stimulation of the NPR-B also results in the production of cGMP.38 However, the NPR-B is expressed in abundance in veins as compared with arteries, in contrast to the NPR-A, which expres¬sion is comparable in both vessel types.44 There¬fore, activation of NPR-B (mostly by CNP) increases cGMP in vascular smooth muscle cells and medi¬ates vasorelaxation thus resulting in a stronger hypotensive effect than NPR-A activation, since the latter also leads to arterial vasoconstriction.45 Knock-out mice models for NPR-B developed dwarfism and female infertility.46 Although stimu¬lation of the NPR-B may not lead to natriuresis or diuresis, recent studies suggest that the NPR-B may play a more important cardioprotective role as was assumed up till now.47 The sensitivity of the NPR-B for the different NPs ranks as follows: CNP > VNP > > ANP = DNP > BNP.3,41,48
NPR-C
The NPR-C is expressed in several organs including lungs, brain, heart, adrenal glands, kidneys, mes- enterium, fat tissue, placenta, veins and aorta. In contrast to NPR-A and NPR-B, it lacks the kinase homology and guanylyl cyclase domains necessary for cGMP production while it is in addition expressed as a dimer. Data derived from a trans¬genic mouse model in which NPR-C was genetically ablated showed that the half-life of radiolabelled ANP in the circulation of homozygote mice lacking NPR-C was 66% longer than in wild-type animals and led to the conclusion that NPR-C functioned mainly as a modulator of NP availability at target organs.49,50
Like in other nutrient-type trans¬membrane receptors, the NPR-C-bound ligand undergoes lysosomal hydrolysis followed by receptor recycling to the cell surface.38 There are however some studies indicating that the NPR-C may mediate anti-proliferative effects of B-type natriuretic peptide and CNP in cardiac fibroblasts.51 The sensitivity of NPR- C for the different NPs ranks as follows: VNP = ANP > CNP > BNP = DNP.3,38,41,51,52
NPR-D
The NPR-D is a unique NPR which has thus far only been isolated in the eel.53 NPR-D is presumed to derive from the NPR-C and lacks the guanylyl cyclase catalytic domain as well. However, in contrast to NPR-D, it acts as a tetramer (like NPR-A and NPR-B). In addition, NPR-D is expressed in limited organs (especially the brain), whereas NPR-C is expressed widely in various organs. However, the present knowledge of the function of the NPR-D is limited. The sensitivity of the NPR-D for NPs in the eel ranks as follows: ANP = VNP > CNP.41
Neutral endopeptidase (NEP)
NEP, also called neprilysin, is a membrane-bound metalloprotease of 87e96 kDa and activators as adenylate cyclase, glucocorticoid, thrombin, calcitonin, and cytokines (such as interleukin-1 b, tumor necrosis factor-alfa and granulocyte— macrophage colony-stimulating factor) can increase NEP mRNA expression and activity.54 Although initially found abundantly in the bush border of the kidney, NEP is also expressed in several other tissues, as for instance in brain and lung.55,56 Interestingly, although the degradation of NPs may be studied most intensively, NEP is also responsible for the inactivation of enkephalins, neuropeptides such as substance P and possibly b-amyloid peptide, the latter highly expressed in Alzheimer’s disease.56
Together with the clearance receptor NPR-C, NEP is responsible for the active elimination from the circulation of NPs via hydrolysis. Parallel to receptor activity, NEP activity also varies for the different NPs, probably due to the fact that a longer C-terminal tail leads to greater resistance for NEP. The rank order of hydrolysis by NEP is CNP > ANP > BNP > DNP, with DNP as the most NEP resistant peptide.57,58
Amino-terminal natriuretic peptide pro-frag¬ments, such as NT-proBNP, are not cleared by either NPRs or NEPs, and is thought to be cleared by organs with high degrees of blood flow, such as the kidney.
Processing of pre-proBNP and its products
As both B-type natriuretic peptide and NT-proBNP are by far the most widely measured natriuretic peptides in medicine, a better understanding of their biology is necessary. The translation of the BNP gene is evoked by several stimuli (see Fig. 4).
The most important stimuli are cardiomyocytal stretch and ischemia/ hypoxia, but other stimuli such as endothelin-1, angiotensin-II, interleukin-1 b and adrenergic agonists, also result in the production of the pre- pro-hormone, namely pre-proBNP1e134.59 This peptide undergoes rapid removal of a 26-amino acid signal peptide, leading to the formation of the 108-amino acid propeptide, proBNP1e108.9 Subse¬quently, proBNPi_i08 is cleaved into two parts by the proteolytic enzymes corin, expressed in the myocardium,60 or furin, ubiquitous distributed in several tissues and in serum61,62: the biologically inert 76-amino acid amino-terminal part NT- proBNP^, and the biologically active 32-amino acid molecule BNP1e32.
Once in circulation, the BNP1e32 molecule is most likely rapidly degraded in fragments that dominate in number relative to mature BNP1e32, but are measured by the commercial BNP tests. Using highly sophisticated analyzing methods, Hawkridge et al. surprisingly found a complete absence of BNP1e32 in the blood of heart failure patients, while commercially available BNP assays tested positive for elevated BNP concentrations.63
Among the more important fragments of BNP that circulate is BNP3_32, which results from the cleavage of B-type natriuretic peptide 1e32 by dipeptidyl peptidase-IV (DPP-IV). Cleavage by DPP-IV seems not to change the molecule’s resistance to further final degra¬dation by neutral endopeptidases (NEP).64 Addi¬tionally, a recent study has demonstrated that the peptidase meprin A, which is highly expressed in the kidney, processes BNP1e32 to BNP7_32 as a necessary step for degradation by NEP; it is however not known yet whether this step also occurs in the degradation of BNP3_32.65
The final elimination of BNP from the circulation is processed via several mechanisms: as discussed previously, BNP is eliminated actively via elimina¬tion by the clearance receptor NPR-C. degradation by NEP and by passive renal filtration. The relative contribution of BNP elimination via NEP and NPR-C is probably equal.66 In addition, BNP is also cleared by the kidneys.67
Figure 4 The cleavage process of pre-proBNP.
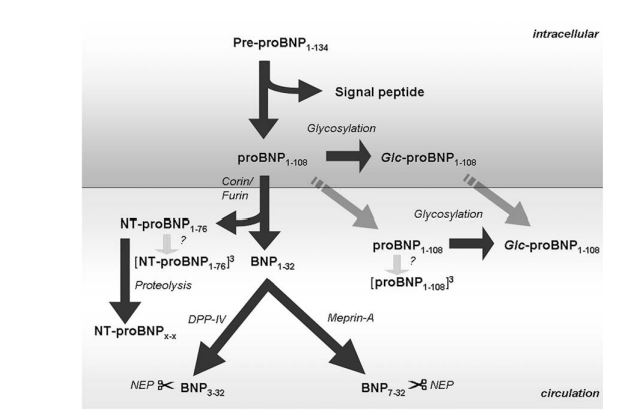
Pre-proBNP = pre-proB-type natriuretic peptide; proBNP = proB-type natriuretic peptide; BNP = B-type natriuretic peptide; DPP-IV = dipeptidyl peptidase-IV; NEP = neutral endopepti- dase; Glc-proBNP = glycosylated proB-type natriuretic peptide.
Because the amino-terminal region of NT- proBNPi_76 (and thus proBNP1e108) contains sequences that permit oligomerization, a trimer of NT-proBNP1e76 and proBNP1e108 has been suggested by some authors, yet this could not be confirmed by others.68,69 Also, fragmentation of NT-proBNP1e76 may exist in vivo because proteol¬ysis can occur at both the carboxy- and amino- terminal ends.70 In addition, NT-proBNP is filtered from the circulation via the kidneys, parallel to BNP.67 Lastly, proBNP1e108 migrates into the circulation as well.71 ProBNP1e108 may then be cleft by furin or may become glycosylated to a variable degree. Glycosylation might take place in the myocardial tissue before leaking in the circulation as well as in the circulation itself.72
BNP and NT-proBNP testing in humans and animals
As mentioned previously, after stimulation of car- diomyocytes in cardiovascular diseases, pre- proBNP production is up-regulated, resulting in an increase of the BNP and NT-proBNP concentrations in the circulation.3 Accordingly, there has been great interest in their use for the diagnosis or exclusion of heart failure, as well as for prognostic and therapeutic applications.
Human applications
In rapid time, both B-type natriuretic peptide and NT-proBNP have emerged as established cardiovascular biomarkers in humans, especially in the syndrome of heart failure.20 Indeed, both peptides have shown to be of excellent value in the diagnosis of acute heart failure in human subjects attending the emergency department for acute dyspnea.73,74 In addition, both peptides strongly predict prognosis in these patients, as well as in acute coronary syndrome or stable ischemic cardiac diseases.75-77 As well, emerging data suggest that pharmacological therapy based on NT-proBNP concentrations with clinically routine therapy, that BNP and NT-proBNP guided treatment was superior.78,79 When used for diagnosis of heart failure in patients with ambiguous symptoms, concentrations of both peptides should not be interpreted blindly but in the context of presentation, since several factors may influence concentrations as depicted in Table 1. Nonetheless, when used in the context of sound clinical judgment and with the broad differ¬ential diagnosis in mind, both BNP and NT-proBNP have been shown to be of value for correctly iden¬tifying or excluding heart failure.73,80
Table 1 Factors associated with increased (NT- pro)BNP concentrations. LV = left ventricle; LVH = left ventricular hypertrophy.
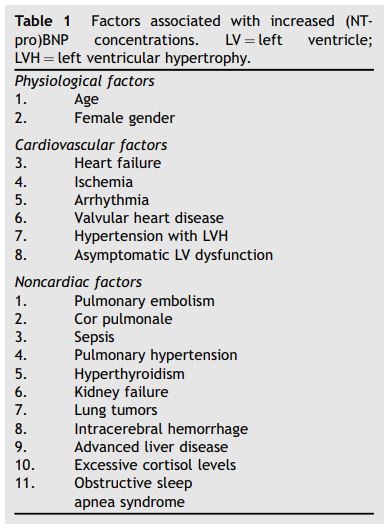
In humans, certain important factors need to be kept in mind when using BNP and NT-proBNP for diagnosis or exclusion of the diagnosis of heart failure. Both increased body mass index (BMI) and impairment in renal function are two factors associated with lower and higher concentrations of BNP and NT-proBNP, respectively, and are worth discussing more into detail.
Obesity, usually objectified in humans as an increased BMI was surprisingly found to be associated with lower BNP levels,81 despite higher left ventricular end diastolic filling pressures in obese patients.82 Although it was first hypothesized that this may be due to an increased NPR-C activity of fat tissue, Krauser et al. showed that obesity was also associated with lower NT-proBNP, which is not cleared by the NPR-C.83 Furthermore, it was shown in human patients undergoing bariatric surgery, that within these patients both BNP as well as NT- proBNP concentrations increased, while BMI decreased.84 Since Das et al. found that the inverse relationship between BNP and NT-proBNP with BMI seems to be mediated by lean mass rather than fat mass, and inverse to BMI-associated changes in androgens.85 It is now hypothesized that a substance produced in the lean mass, possibly androgens, suppresses either synthesis or release of pre-proBNP from cardiomyocytes.85 Clinically, the effect of rising BMI on BNP and NT-proBNP is a generally lower sensitivity of the markers, with perhaps a more significant effect on BNP: authors suggest a lower BNP cut-point of 54 pg/mL in obese humans,86 while for NT-proBNP lower values were not accompanied by the need for a change in cut- points to see similar performance.87
On the other hand, renal disease is typically associated with higher than expected B-type natriuretic peptide and NT- proBNP values. Since a moderate yet consistent correlation was seen between renal impairment and elevated BNP and NT-proBNP concentrations, it has been questioned whether this association is based on a decrease in renal clearance of these molecules, or whether this association reflects the strong interaction between cardiac and renal (dys)function, also called the 'cardio-renal syndrome’.88 Furthermore, since BNP tends to increase less than NT-proBNP in renal impairment,89 there has been an intense debate whether concentrations of BNP (also cleared by NEP and NPR-C) are less affected by renal impairment.90 Yet, observational head-to-head comparisons showed that both molecules performed equally, also and especially in the presence of renal impairment.91-93 In addition, mechanistic studies comparing the renal clearance of both molecules, also showed no differences in renal handling of the molecules.67
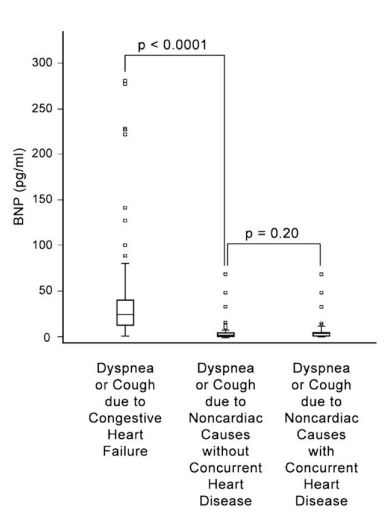
Figure 5 Box plot of plasma B-type natriuretic peptide (BNP) concentrations for 3 groups of dogs presenting with cough or dyspnea. Natriuretic peptides were useful for correct diagnosis of heart failure in this context. Derived from DeFrancesco et al.105
Both B-type natriuretic peptide and NT-proBNP have shown to be excellent predictors of prognosis, in the short term as well as in the longer term, and not only in patients with acute or chronic heart failure but in the complete spectrum of cardiovascular dis- eases.75,80,94-96 Interestingly, a decrease in NT- proBNP concentrations was also associated with improved outcome,97 and smaller studies even suggest that natriuretic peptide guided treatment may enable clinicians to reduce morbidity and mortality in heart failure patients in the out¬patient setting.78,79 However, further studies are clearly needed in order to delineate the ideal population, as well as to elucidate the proper therapeutic strategy, since it is for instance not known what the (absolute or relative) target B-type natriuretic peptide or NT-proBNP concentration should be.
Natriuretic peptide testing in veterinary medicine
NP testing has now found its way into veterinarian practice. As in humans, the first veterinary experi¬ences were with ANP testing and were promising: it was found that higher concentrations of ANP were associated with mitral valvular disease, heartworm disease and heart failure in dogs.98-100 However, while in humans BNP and NT-proBNP testing was found to be superior to ANP testing, in veterinarian practice a different picture has emerged. Early use of human BNP assays in dogs returned inferior results to ANP.101 This may be due to the fact that canine pre-proBNP only shares 45% homology with human pre-proBNP (limiting ability to use human BNP assays in this context); in contrast, canine pre-proANP has 87% in common with the human variant.102,103 In addition, canine BNP only has a half life of 90s, which makes testing for it more challenging.104
However, with the development of canine- specific BNP assays, progress has been made. DeFrancesco et al. demonstrated, among 330 dogs presenting with cough or dyspnea, that, although serum BNP concentrations were remarkably lower than in humans, BNP was able to discriminate between heart failure and non-heart failure as a cause for symptoms (even in dogs with a previous medical history of heart failure) and also 7 correlated with severity of heart failure.105 This is also depicted in Fig. 5.
These findings were confirmed by Oyama et al. who demonstrated similar results for NT-proBNP testing in dogs.106 However, unlike in humans, given the frailties of BNP, in animal applications, ANP and its cleavage equivalent NT-proANP remained the subject of study. In an interesting head-to-head comparison in 48 dogs suffering from respiratory distress, Prosek et al. showed that a human NT-proANP assay was diagnostically superior to a canine- specific BNP assay.107
In contrast to the weakness of BNP in dogs, NT- proBNP has shown to be an excellent canine cardiovascular biomarker. For example, in a pres¬sure-overload model of aortic banding in 6 male Beagles, it was shown that NT-proBNP better correlated with left ventricular end diastolic pressures when compared to NT-proANP.108 In addition, recent publications demonstrated that the diagnostic and prognostic value of NT-proBNP in cats and dogs was superior to NT-proANP.109,110 In contrast to humans,111 B-type natriuretic peptide and NT-proBNP concentrations in animals seem to be less associ¬ated with age or gender, while the associations with body weight and renal impairment have been found.105,106,110,112
While application of NT-proBNP testing for diagnostic evaluation of the dyspneic small animal appears promising—and perhaps bound for stan¬dard practice—further studies are now necessary in order to optimally implement such testing in daily small animal practice. More data are needed regarding the prognostic value of NT-proBNP in small animals as well, and potential for NT-proBNP guided therapeutic intervention in such patients needs exploration. Furthermore, since ANP was not found to be diagnostic in horses with valvular disease and equine (and other) BNP and NT-proBNP are not available, this area is still ill-explored in larger animals.113
References
- de Bold AJ, Borenstein HB, Veress AT, Sonnenberg H. A rapid and potent natriuretic response to intravenous injection of atrial myocardial extracts in rats. Life Sci 1981;28:89-94.
- Kangawa K, Fukuda A, Kubota I, Hayashi Y, Minamitake Y, Matsuo H. Human atrial natriuretic polypeptides (hANP): purification, structure synthesis and biological activity. J Hypertens Suppl 1984;2:S321-S323.
- Levin ER, Gardner DG, Samson WK. Natriuretic peptides. N Engl J Med 1998;339:321-328.
- Takei Y, Ogoshi M, Inoue K. A 'reverse' phylogenetic approach for identification of novel osmoregulatory and cardiovascular hormones in vertebrates. Front Neuro- endocrinol 2007;28:143-160.
- Vesely DL. Natriuretic peptides and acute renal failure. Am J Physiol Renal Physiol 2003;285:F167-F177.
- Saito Y, Nakao K, Arai H, Nishimura K, Okumura K, Obata K, Takemura G, Fujiwara H, Sugawara A, Yamada T, Itoh H, Mukoyama M, Hosoda K, Kawai C, Ban T, Yasue H, Imura H. Augmented expression of atrial natriuretic polypeptide gene in ventricle of human failing heart. J Clin Invest 1989; 83:298-305.
- Gunning M, Brenner BM. Urodilatin: a potent natriuretic peptide of renal origin. CurrOpin Nephrol Hypertens 1993; 2:857-862.
- Zeidel ML. Regulation of collecting duct Na+ reabsorption byANP 31-67. Clin Exp Pharmacol Physiol 1995;22:121-124.
- Sudoh T, Kangawa K, Minamino N, Matsuo H. A new natri¬uretic peptide in porcine brain. Nature 1988;332:78-81.
- Porter JG, Arfsten A, Palisi T, Scarborough RM, Lewicki JA, Seilhamer JJ. Cloning of a cDNA encoding porcine brain natriuretic peptide. J Biol Chem 1989;264:6689-6692.
- Yamamoto K, Burnett Jr JC, Jougasaki M, Nishimura RA, Bailey KR, Saito Y, Nakao K, Redfield MM. Superiority of brain natriuretic peptide as a hormonal marker of ventricular systolic and diastolic dysfunction and ventric¬ular hypertrophy. Hypertension 1996;28:988-994.
- Clerico A, Iervasi G, Del Chicca MG, Emdin M, Maffei S, Nannipieri M, Sabatino L, Forini F, Manfredi C, Donato L. Circulating levels of cardiac natriuretic peptides (ANP and BNP) measured by highly sensitive and specific immunor- adiometric assays in normal subjects and in patients with different degrees of heart failure. J Endocrinol Invest 1998;21:170-179.
- Nakagawa O, Ogawa Y, Itoh H, Suga S, Komatsu Y, Kishimoto I, Nishino K, Yoshimasa T, Nakao K. Rapid tran¬scriptional activation and early mRNA turnover of brain natriuretic peptide in cardiocyte hypertrophy. Evidence for brain natriuretic peptide as an ''emergency'' cardiac hormone against ventricular overload. J Clin Invest 1995; 96:1280-1287.
- Goetze JP, Gore A, MollerCH, Steinbruchel DA, Rehfeld JF, Nielsen LB. Acute myocardial hypoxia increases BNP gene expression. FASEB J 2004;18:1928-1930.
- Brunner-La Rocca HP, Kaye DM, Woods RL, Hastings J, Esler MD. Effects of intravenous brain natriuretic peptide on regional sympathetic activity in patients with chronic heart failure as compared with healthy control subjects. J Am Coll Cardiol 2001;37:1221-1227.
- Clarkson PB, Wheeldon NM, MacFadyen RJ, Pringle SD, MacDonald TM. Effects of brain natriuretic peptide on exercise hemodynamics and neurohor¬mones in isolated diastolic heart failure. Circulation 1996;93:2037-2042.
- van der Zander K, Houben AJ, Kroon AA, De Mey JG, Smits PA, de Leeuw PW. Nitric oxide and potassium chan¬nels are involved in brain natriuretic peptide induced vasodilatation in man. J Hypertens 2002;20:493-499.
- Cao L, Gardner DG. Natriuretic peptides inhibit DNA synthesis in cardiac fibroblasts. Hypertension 1995;25: 227-234.
- Fujisaki H, Ito H, Hirata Y, Tanaka M, Hata M, Lin M, Adachi S, Akimoto H, Marumo F, Hiroe M. Natriuretic peptides inhibit angiotensin II-induced proliferation of rat cardiac fibroblasts by blocking endothelin-1 gene expres¬sion. J Clin Invest 1995;96:1059-1065.
- Braunwald E. Biomarkers in heart failure. N Engl J Med 2008;358:2148-2159.
- Horio T, Tokudome T, Maki T, Yoshihara F, Suga S, Nishikimi T, Kojima M, Kawano Y, Kangawa K. Gene expression, secretion, and autocrine action of C-type natriuretic peptide in cultured adult rat cardiac fibro¬blasts. Endocrinology 2003;144:2279-2284.
- Wei CM, Heublein DM, Perrella MA, Lerman A, Rodeheffer RJ, McGregor CG, Edwards WD, Schaff HV, Burnett Jr JC. Natriuretic peptide system in human heart failure. Circulation 1993;88:1004-1009.
- Del Ry S, Passino C, Emdin M, Giannessi D. C-type natri¬uretic peptide and heart failure. Pharmacol Res 2006;54: 326-333.
- Barr CS, Rhodes P, Struthers AD. C-type natriuretic peptide. Peptides 1996;17:1243-1251.
- Igaki T, Itoh H, Suga SI, Hama N, Ogawa Y, Komatsu Y, Yamashita J, Doi K, Chun TH, Nakao K. Effects of intra¬venously administered C-type natriuretic peptide in humans: comparison with atrial natriuretic peptide. Hypertens Res 1998;21:7-13.
- Carvajal JA, Aguan K, Thompson LP, Buhimschi IA, Weiner CP. Natriuretic peptide-induced relaxation of myometrium from the pregnant guinea pig is not mediated by guanylate cyclase activation. J Pharmacol Exp Ther 2001;297:181-188.
- El-Gehani F, Tena-Sempere M, Ruskoaho H, Huhtaniemi I. Natriuretic peptides stimulate steroidogenesis in the fetal rat testis. Biol Reprod 2001;65:595-600.
- Walther T, Stepan H. C-type natriuretic peptide in repro¬duction, pregnancy and fetal development. J Endocrinol 2004;180:17-22.
- Schweitz H, Vigne P, Moinier D, Frelin C, Lazdunski M. A new member of the natriuretic peptide family is present in the venom of the green mamba (Dendroaspis angusticeps). J Biol Chem 1992;267:13928-13932.
- Singh G, Kuc RE, Maguire JJ, Fidock M, Davenport AP. Novel snake venom ligand dendroaspis natriuretic peptide is selective for natriuretic peptide receptor-A in human heart: downregulation of natriuretic peptide receptor-Ain heart failure. Circ Res 2006;99:183-190.
- Schirger JA, Heublein DM, Chen HH, Lisy O, Jougasaki M, Wennberg PW, Burnett Jr JC. Presence of dendroaspis natriuretic peptide-like immunoreactivity in human plasma and its increase during human heart failure. Mayo Clin Proc 1999;74:126-130.
- Lisy O, Lainchbury JG, Leskinen H, Burnett Jr JC. Thera¬peutic actions of a new synthetic vasoactive and natri¬uretic peptide, dendroaspis natriuretic peptide, in experimental severe congestive heart failure. Hyperten¬sion 2001;37:1089-1094.
- Carroll RL. Vertebrate paleontology and evolution. New York: Freeman, W.H and Company; 1998. 16-25.
- Trajanovska S, Inoue K, Takei Y, Donald JA. Genomic analyses and cloning of novel chicken natriuretic peptide genes reveal new insights into natriuretic peptide evolu¬tion. Peptides 2007;28:2155-2163.
- Inoue K, Naruse K, Yamagami S, Mitani H, Suzuki N, Takei Y. Four functionally distinct C-type natriuretic peptides found in fish reveal evolutionary history of the natriuretic peptide system. Proc Natl Acad Sci U S A 2003; 100:10079-10084.
- Inoue K, Sakamoto T, Yuge S, Iwatani H, Yamagami S, Tsutsumi M, Hori H, Cerra MC, Tota B, Suzuki N, Okamoto N, Takei Y. Structural and functional evolution of three cardiac natriuretic peptides. Mol Biol Evol 2005;22: 2428-2434.
- Pandey KN. Biology of natriuretic peptides and their receptors. Peptides 2005;26:901-932.
- Potter LR, Abbey-Hosch S, Dickey DM. Natriuretic peptides, their receptors, and cyclic guanosine mono¬phosphate-dependent signaling functions. Endocr Rev 2006;27:47-72.
- Lee CY, Burnett Jr JC. Natriuretic peptides and thera¬peutic applications. Heart Fail Rev 2007;12:131-142.
- Lopez MJ, Wong SK, Kishimoto I, Dubois S, Mach V, Friesen J, Garbers DL, Beuve A. Salt-resistant hypertension in mice lacking the guanylyl cyclase-A receptor for atrial natriuretic peptide. Nature 1995;378:65-68.
- Inoue K, Takei Y. Molecular evolution of the natriuretic peptide system as revealed by comparative genomics. Comp Biochem Physiol Part D 2006;1:69-76.
- Best PJ, Burnett JC, Wilson SH, Holmes Jr DR, Lerman A. Dendroaspis natriuretic peptide relaxes isolated human arteries and veins. Cardiovasc Res 2002;55:375-384.
- Takei Y, Hirose S. The natriuretic peptide system in eels: a key endocrine system for euryhalinity? Am J Physiol Regul Integr Comp Physiol 2002;282:R940-R951.
- Wei CM, Aarhus LL, Miller VM, Burnett Jr JC. Action of C- type natriuretic peptide in isolated canine arteries and veins. Am J Physiol 1993;264:H71-H73.
- Wennberg PW, Miller VM, Rabelink T, Burnett Jr JC. Further attenuation of endothelium-dependent relaxation imparted by natriuretic peptide receptor antagonism. Am J Physiol 1999;277:H1618-H1621.
- Tamura N, Doolittle LK, Hammer RE, Shelton JM, Richardson JA, Garbers DL. Critical roles of the guanylyl cyclase B receptor in endochondral ossification and development of female reproductive organs. Proc Natl Acad Sci U S A 2004;101:17300-17305.
- Dickey DM, Flora DR, Bryan PM, Xu X, Chen Y, Potter LR. Differential regulation of membrane guanylyl cyclases in congestive heart failure: natriuretic peptide receptor (NPR)-B, not NPR-A, is the predominant natriuretic peptide receptor in the failing heart. Endocrinology 2007; 148:3518-3522.
- Piao FL, Park SH, Han JH, Cao C, Kim SZ, Kim SH. Den¬droaspis natriuretic peptide and its functions in pig ovarian granulosa cells. Regul Pept 2004;118:193-198.
- Jaubert J, Jaubert F, Martin N, Washburn LL, Lee BK, Eicher EM, Guenet JL. Three new allelic mouse mutations that cause skeletal overgrowth involve the natriuretic peptide receptor C gene (Npr3). Proc Natl Acad Sci U S A 1999;96:10278-10283.
- Matsukawa N, GrzesikWJ, Takahashi N, Pandey KN, PangS, Yamauchi M, Smithies O. The natriuretic peptide clearance receptor locally modulates the physiological effects of the natriuretic peptide system. Proc Natl Acad Sci U S A 1999; 96:7403-7408.
- Rose RA, Giles WR. Natriuretic peptide C receptor signal¬ling in the heart and vasculature. J Physiol 2008;586: 353-366.
- Johns DG, Ao Z, Heidrich BJ, Hunsberger GE, Graham T, Payne L, Elshourbagy N, Lu Q, Aiyar N, Douglas SA. Den- droaspis natriuretic peptide binds to the natriuretic peptide clearance receptor. Biochem Biophys Res Commun 2007;358:145-149.
- Kashiwagi M, Katafuchi T, Kato A, Inuyama H, Ito T, Hagiwara H, Takei Y, Hirose S. Cloning and properties of a novel natriuretic peptide receptor, NPR-D. Eur J Biochem 1995;233:102-109.
- Roques BP, Noble F, Dauge V, Fournie-Zaluski MC, Beaumont A. Neutral endopeptidase 24.11: structure, inhibition, and experimental and clinical pharmacology. Pharmacol Rev 1993;45:87-146.
- Kerr MA, Kenny AJ. The purification and specificity of a neutral endopeptidase from rabbit kidney brush border. Biochem J 1974;137:477-488.
- Turner AJ, Isaac RE, Coates D. The neprilysin (NEP) family of zinc metalloendopeptidases: genomics and function. Bioessays 2001;23:261-269.
- Chen HH, Lainchbury JG, Burnett Jr JC. Natriuretic peptide receptors and neutral endopeptidase in mediating the renal actions of a new therapeutic synthetic natri¬uretic peptide dendroaspis natriuretic peptide. J Am Coll Cardiol 2002;40:1186-1191.
- Dussaule JC, Stefanski A, Bea ML, Ronco P, Ardaillou R. Characterization of neutral endopeptidase in vascular smooth muscle cells of rabbit renal cortex. Am J Physiol 1993;264:F45-F52.
- Martinez-RumayorA, RichardsAM, Burnett JC, Januzzi Jr JL. Biology of the natriuretic peptides. Am J Cardiol 2008;101: 3-8.
- Yan W, Wu F, Morser J, Wu Q. Corin, a transmembrane cardiac serine protease, acts as a pro-atrial natriuretic peptide-converting enzyme. Proc Natl Acad Sci U S A 2000; 97:8525-8529.
- Sawada Y, Suda M, Yokoyama H, Kanda T, Sakamaki T, Tanaka S, Nagai R, Abe S, Takeuchi T. Stretch-induced hypertrophic growth of cardiocytes and processing of brain-type natriuretic peptide are controlled by propro¬tein-processing endoprotease furin. J Biol Chem 1997;272: 20545-20554.
- Hunt PJ, Espiner EA, Nicholls MG, Richards AM, Yandle TG. The role of the circulation in processing pro-brain natri¬uretic peptide (proBNP) to amino-terminal BNP and BNP- 32. Peptides 1997;18:1475-1481.
- Hawkridge AM, Heublein DM, Bergen 3rd HR, Cataliotti A, Burnett Jr JC, Muddiman DC. Quantitative mass spectral evidence for the absence of circulating brain natriuretic peptide (BNP-32) in severe human heart failure. Proc Natl Acad Sci U S A 2005;102:17442-17447.
- Brandt I, Lambeir AM, Ketelslegers JM, Vanderheyden M, Scharpe S, De Meester I. Dipeptidyl-peptidase IV converts intact B-type natriuretic peptide into its des-SerPro form. Clin Chem 2006;52:82-87.
- Pankow K, Wang Y, Gembardt F, Krause E, Sun X, Krause G, Schultheiss HP, Siems WE, Walther T. Successive action of meprin A and neprilysin catabolizes B-type natriuretic peptide. Circ Res 2007;101:875-882.
- Smith MW, Espiner EA, Yandle TG, Charles CJ, RichardsAM. Delayed metabolism of human brain natriuretic peptide reflects resistance to neutral endopeptidase. J Endocrinol 2000;167:239-246.
- Goetze JP, Jensen G, Moller S, Bendtsen F, Rehfeld JF, Henriksen JH. BNP and N-terminal proBNP are both extrac¬ted in the normal kidney. Eur J Clin Invest 2006;36:8-15.
- Seidler T, Pemberton C, Yandle T, Espiner E, Nicholls G, Richards M. The amino terminal regions of proBNP and proANP oligomerise through leucine zipper-like coiled-coil motifs. Biochem Biophys Res Commun 1999;255:495-501.
- Crimmins DL. Human N-terminal proBNP is a monomer. Clin Chem 2005;51:1035-1038.
- Ala-Kopsala M, Magga J, Peuhkurinen K, Leipala J, Ruskoaho H, Leppaluoto J, Vuolteenaho O. Molecular heterogeneity has a major impact on the measurement of circulating N-terminal fragments of A- and B-type natri¬uretic peptides. Clin Chem 2004;50:1576-1588.
- Waldo SW, Beede J, Isakson S, Villard-Saussine S, Fareh J, Clopton P, Fitzgerald RL, Maisel AS. Pro-B-type natriuretic peptide levels in acute decompensated heart failure. J Am Coll Cardiol 2008;51:1874-1882.
- Schellenberger U, O'Rear J, Guzzetta A, Jue RA, Protter AA, Pollitt NS. The precursor to B-type natriuretic peptide is an O-linked glycoprotein. Arch Biochem Biophys 2006;451:160-166.
- Maisel AS, Krishnaswamy P, Nowak RM, McCord J, Hollander JE, Duc P, Omland T, Storrow AB, Abraham WT, Wu AH, Clopton P, Steg PG, Westheim A, Knudsen CW, Perez A, Kazanegra R, Herrmann HC, McCullough PA. Rapid measurement of B-type natriuretic peptide in the emergency diagnosis of heart failure. N Engl J Med 2002;347:161-167.
- Januzzi Jr JL, Camargo CA, Anwaruddin S, Baggish AL, Chen AA, Krauser DG, Tung R, Cameron R, Nagurney JT, Chae CU, Lloyd-Jones DM, Brown DF, Foran-Melanson S, Sluss PM, Lee-Lewandrowski E, Lewandrowski KB. The N- terminal Pro-BNP Investigation of Dyspnea in the Emergency Department (PRIDE) Study. Am J Cardiol 2005;95:948-954.
- Januzzi Jr JL, Sakhuja R, O'Donoghue M, Baggish AL, Anwaruddin S, Chae CU, Cameron R, Krauser DG, Tung R, Camargo Jr CA, Lloyd-Jones DM. Utility of amino-terminal pro-brain natriuretic peptide testing for prediction of 1-year mortality in patients with dyspnea treated in the emergency department. Arch Intern Med 2006;166: 315-320.
- Omland T, Persson A, Ng L, O'Brien R, Karlsson T, Herlitz J, Hartford M, Caidahl K. N-terminal pro-B-type natriuretic peptide and long-term mortality in acute coronary syndromes. Circulation 2002;106:2913-2918.
- Omland T, Richards AM, Wergeland R, Vik-Mo H. B-type natriuretic peptide and long-term survival in patients with stable coronary artery disease. Am J Cardiol 2005;95: 24-28.
- Troughton RW, Frampton CM, Yandle TG, Espiner EA, Nicholls MG, Richards AM. Treatment of heart failure guided by plasma aminoterminal brain natriuretic peptide (N-BNP) concentrations. Lancet 2000;355:1126-1130.
- Jourdain P, Jondeau G, Funck F, Gueffet P, Le Helloco A, Donal E, Aupetit JF, Aumont MC, Galinier M, Eicher JC, Cohen-Solal A, Juilliere Y. Plasma brain natriuretic peptide-guided therapy to improve outcome in heart failure: the STARS-BNP Multicenter Study. J Am Coll Car¬diol 2007;49:1733-1739.
- Januzzi Jr JL, van Kimmenade RRJ, Lainchbury JG, Bayes- Genis A, Pinto YM, Richards AM. NT-proBNP testing for diagnosis and short-term prognosis in acute congestive heart failure: an international pooled analysis of 1256 patients. The International Collaborative of NT-proBNP (ICON) Study. Eur Heart J 2006;27:330-337.
- Horwich TB, Fonarow GC, Hamilton MA, MacLellan WR, Woo MA, Tillisch JH. The relationship between obesity and mortality in patients with heart failure. J Am Coll Cardiol 2001;38:789-795.
- Taylor JA, Christenson RH, Rao K, Jorge M, Gottlieb SS. B-type natriuretic peptide and N-terminal pro B-type natriuretic peptide are depressed in obesity despite higher left ventricular end diastolic pressures. Am Heart J 2006; 152:1071-1076.
- Krauser DG, Lloyd-Jones DM, Chae CU, Cameron R, Anwaruddin S, Baggish AL, Chen AA, Tung R, Januzzi Jr JL. Effect of body mass index on natriuretic peptide levels in patients with acute congestive heart failure: a ProBNP investigation of dyspnea in the emergency department (PRIDE) substudy. Am Heart J 2005;149:744-750.
- van Kimmenade R, van Dielen F, Bakker J, Nijhuis J, Crijns H, Buurman W, van Dieijen-Visser M, Greve JW, Pinto Y. Is brain natriuretic peptide production decreased in obese subjects? J Am Coll Cardiol 2006;47:886-887.
- Das SR, Drazner MH, Dries DL, Vega GL, Stanek HG, Abdullah SM, Canham RM, Chung AK, Leonard D, Wians Jr FH, de Lemos JA. Impact of body mass and body composition on circulating levels of natriuretic peptides: results from the Dallas Heart Study. Circulation 2005;112: 2163-2168.
- Daniels LB, Clopton P, Bhalla V, Krishnaswamy P, Nowak RM, McCord J, Hollander JE, Duc P, Omland T, Storrow AB, Abraham WT, Wu AH, Steg PG, Westheim A, Knudsen CW, Perez A, Kazanegra R, Herrmann HC, McCullough PA, Maisel AS. How obesity affects the cut- points for B-type natriuretic peptide in the diagnosis of acute heart failure. Results from the Breathing Not Prop¬erly Multinational Study. Am Heart J 2006;151:999-1005.
- Bayes-Genis A, Lloyd-Jones DM, van Kimmenade RR, Lainchbury JG, Richards AM, Ordonez-Llanos J, Santalo M, Pinto YM, Januzzi Jr JL. Effect of body mass index on diagnostic and prognostic usefulness of amino-terminal pro-brain natriuretic peptide in patients with acute dysp¬nea. Arch Intern Med 2007;167:400-407.
- Stevenson LW, Nohria A, Mielniczuk L. Torrent or torment from the tubules? Challenge of the cardiorenal connec¬tions. J Am Coll Cardiol 2005;45:2004-2007.
- Kemperman H, van den Berg M, Kirkels H, de Jonge N. B-type natriuretic peptide (BNP) and N-terminal proBNP in patients with end-stage heart failure supported by a left ventricular assist device. Clin Chem 2004;50:1670-1672.
- McCullough PA, Omland T, Maisel AS. B-type natriuretic peptides: a diagnostic breakthrough for clinicians. Rev Cardiovasc Med 2003;4:72-80.
- Richards AM, Nicholls MG, Espiner EA, Lainchbury JG, Troughton RW, Elliott J, Frampton C, Crozier IG, Yandle T, Doughty R, MacMahon S, Sharpe N. Comparison of B-type natriuretic peptides for assessment of cardiac function and prognosis in stable ischemic heart disease. J Am Coll Cardiol 2006;47:52-60.
- Austin WJ, Bhalla V, Hernandez-Arce I, Isakson SR, Beede J, Clopton P, Maisel AS, Fitzgerald RL. Correlation and prognostic utility of B-type natriuretic peptide and its amino-terminal fragment in patients with chronic kidney disease. Am J Clin Pathol 2006;126:506-512.
- McKie PM, Rodeheffer RJ, Cataliotti A, Martin FL, Urban LH, Mahoney DW, Jacobsen SJ, Redfield MM, Burnett Jr JC. Amino-terminal pro-B-type natriuretic peptide and B-type natriuretic peptide. biomarkers for mortality in a large community-based cohort free of heart failure. Hypertension 2006;47:874-880.
- Omland T, de Lemos JA, Morrow DA, Antman EM, Cannon CP, Hall C, Braunwald E. Prognostic value of N- terminal pro-atrial and pro-brain natriuretic peptide in patients with acute coronary syndromes. Am J Cardiol 2002;89:463-465.
- Harrison A, Morrison LK, Krishnaswamy P, Kazanegra R, Clopton P, Dao Q, Hlavin P, Maisel AS. B-type natriuretic peptide predicts future cardiac events in patients presenting to the emergency department with dyspnea. Ann EmergMed 2002;39:131-138.
- Kragelund C, Gronning B, Kober L, Hildebrandt P, Steffensen R. N-terminal pro-B-type natriuretic peptide and long-term mortality in stable coronary heart disease. N Engl J Med 2005;352:666-675.
- Bettencourt P, Azevedo A, Pimenta J, Frioes F, Ferreira S, Ferreira A. N-terminal-pro-brain natriuretic peptide predicts outcome after hospital discharge in heart failure patients. Circulation 2004;110:2168-2174.
- Haggstrom J, Hansson K, Karlberg BE, Kvart C, Olsson K. Plasma concentration of atrial natriuretic peptide in relation to severity of mitral regurgitation in Cavalier King Charles Spaniels. Am J Vet Res 1994;55:698-703.
- Takemura N, Koyama H, Sako T, Ando K, Motoyoshi S, Marumo F. Plasma atrial natriuretic peptide (ANP) in dogs with mild heartworm infection. J Vet Med Sci 1991;53: 323-325.
- VollmarAM, Reusch C, Kraft W, Schulz R. Atrial natriuretic peptide concentration in dogs with congestive heart failure, chronic renal failure, and hyperadrenocorticism. Am J Vet Res 1991;52:1831-1834.
- Asano K, Masuda K, Okumura M, Kadosawa T, Fujinaga T. Plasma atrial and brain natriuretic peptide levels in dogs with congestive heart failure. J Vet Med Sci 1999;61: 523-529.
- Liu ZL, Wiedmeyer CE, Sisson DD, Solter PF. Cloning and characterization of feline brain natriuretic peptide. Gene 2002;292:183-190.
- Oikawa S, Imai M, Inuzuka C, Tawaragi Y, Nakazato H, Matsuo H. Structure of dog and rabbit precursors of atrial natriuretic polypeptides deduced from nucleotide sequence of cloned cDNA. Biochem Biophys Res Commun 1985;132:892-899.
- Thomas CJ, Woods RL. Haemodynamic action of B-type natriuretic peptide substantially outlasts its plasma half¬life in conscious dogs. Clin Exp Pharmacol Physiol 2003;30: 369-375.
- DeFrancesco TC, Rush JE, Rozanski EA, Hansen BD, Keene BW, Moore DT, Atkins CE. Prospective clinical evaluation of an ELISA B-type natriuretic peptide assay in the diagnosis of congestive heart failure in dogs pre¬senting with cough or dyspnea. J Vet Intern Med 2007;21: 243-250.
- OyamaMA, Fox PR, Rush JE, Rozanski EA, Lesser M. Clinical utility of serum N-terminal pro-B-type natriuretic peptide concentration for identifying cardiac disease in dogs and assessing disease severity. J Am Vet Med Assoc 2008;232: 1496-1503.
- Prosek R, Sisson DD, Oyama MA, Solter PF. Distinguishing cardiac and noncardiac dyspnea in 48 dogs using plasma atrial natriuretic factor, B-type natriuretic factor, endo- thelin, and cardiac troponin-I. J Vet Intern Med 2007;21: 238-242.
- Hori Y, Tsubaki M, Katou A, Ono Y, Yonezawa T, Li X, Higuchi SI. Evaluation of NT-Pro BNP and CT-ANP as markers of concentric hypertrophy in dogs with a model of compensated aortic stenosis. J Vet Intern Med 2008;22: 1118-1123.
- Connolly DJ, Magalhaes RJ, Syme HM, Boswood A, Fuentes VL, Chu L, Metcalf M. Circulating natriuretic peptides in cats with heart disease. J Vet Intern Med 2008; 22:96-105.
- Tarnow I, Olsen LH, Kvart C, Hoglund K, Moesgaard SG, Kamstrup TS, Pedersen HD, Haggstrom J. Predictive value of natriuretic peptides in dogs with mitral valve disease. Vet J, in press. The evolution of the natriuretic peptides S21
- Redfield MM, Rodeheffer RJ, Jacobsen SJ, Mahoney DW, Bailey KR, Burnett Jr JC. Plasma brain natriuretic peptide concentration: impact of age and gender. J Am Coll Car¬diol 2002;40:976-982.
- Boswood A, Dukes-McEwan J, Loureiro J, James RA, Martin M, Stafford-Johnson M, Smith P, Little C, Attree S. The diagnostic accuracy of different natriuretic peptides in the investigation of canine cardiac disease. J Small Anim Pract 2008;49:26-32.
- Gehlen H, Sundermann T, Rohn K, Stadler P. Plasma atrial natriuretic peptide concentration in warmblood horses with heart valve regurgitations. J Vet Cardiol 2007;9:99-101.
^Наверх









