Anatomic regurgitant orifice area obtained using 3D-echocardiography as an indicator of severity of mitral regurgitation in dogs with myxomatous mitral valve disease

Author information
Müller S., Menciotti G., Borgarelli M. Anatomic regurgitant orifice area obtained using 3D-echocardiography as an indicator of severity of mitral regurgitation in dogs with myxomatous mitral valve disease // J Vet Cardiol. 2017 Oct;19(5):433-440.
Abstract
OBJECTIVES: To determine feasibility and repeatability of measuring the anatomic regurgitant orifice area (AROA) using real-time three-dimensional transthoracic echocardiography (RT3DE) in dogs with myxomatous mitral valve disease (MMVD), and to investigate differences in the AROA of dogs with different disease severity and in different American College of Veterinary Internal Medicine (ACVIM) stages.
ANIMALS: Sixty privately-owned dogs diagnosed with MMVD.
METHODS: The echocardiographic database of our institution was retrospectively searched for dogs diagnosed with MMVD and RT3DE data set acquisition. Dogs were classified into mild, moderate, or severe MMVD according to a Mitral Regurgitation Severity Score (MRSS), and into stage B1, B2 or C according to ACVIM staging. The real-time three-dimensional transthoracic echocardiography (RT3DE) data sets were imported into dedicated software and a short axis plane crossing the regurgitant orifice was used to measure the AROA. Feasibility, inter- and intra-observer variability of measuring the AROA was calculated. Differences in the AROA between dogs in different MRSS and ACVIM stages were investigated.
RESULTS: The AROA was measurable in 60 data sets of 81 selected to be included in the study (74%). The inter- and intra-observer coefficients of variation were 26% and 21%, respectively. The AROA was significantly greater in dogs with a severe MRSS compared with dogs with mild MRSS (p=0.045). There was no difference between the AROA of dogs in different ACVIM clinical stages.
CONCLUSIONS: Obtaining the AROA using real-time three-dimensional transthoracic echocardiography (RT3DE) is feasible and might provide additional information to stratify mitral regurgitation severity in dogs with MMVD. Diagnostic and prognostic utility of the AROA deserves further investigation.
Abbreviations
- ACVIM American College of Veterinary Internal Medicine
- AROA anatomic regurgitant orifice area
- BW body weight
- %CV coefficient of variation
- EROA effective regurgitant orifice area
- LA left atrial
- LA:Ao left atrial to aortic root ratio
- LA Vol/BW left atrial volume indexed to body weight
- MMVD myxomatous mitral valve disease
- MR mitral regurgitation
- MRSS mitral regurgitation severity score
- MV mitral valve
- RT3DE real-time three-dimensional transthoracic echocardiography
- 2DE two-dimensional echocardiography
Introduction
Myxomatous mitral valve disease (MMVD) is the most common acquired cardiac disease in dogs, and the most common cause of congestive heart failure in this species [1,2]. Current classification of the severity of MMVD in dogs is based on both clinical signs and identification of cardiac remodeling by radiographs and echocardiography [3]. In humans, classification of the severity of MMVD is mainly based on echocardiographic criteria [4] with particular attention posed on quantification of mitral regurgitation (MR) [5,6]. In fact, in humans, the severity of MR is associated with prognosis in patients with MMVD [5,7]. It is unclear whether the severity of MR is an independent predictor of survival in dogs with MMVD; however, several studies indirectly suggest that this is also true in this species [8—10].
One of the main factors affecting MR severity is the size of the orifice through which MR occurs[11,12]. Cardiac magnetic resonance imaging is considered the gold standard to assess the mitral regurgitant orifice, but this technique has several limitations in veterinary medicine including requirement for general anesthesia, time, and specific personnel and equipment, therefore resulting in elevated costs and poor applicability in clinical settings. Two-dimensional echocardiography (2DE), in combination with Doppler-based techniques is currently the method of choice for non-invasive evaluation of MR in both humans and dogs [4,9,11,13]. However, obtaining information about the size of the orifice using 2DE and Doppler techniques could be time-consuming, and is demonstrated to suffer from limitations that could arise from misalignment of the scanning plane with the regurgitant jet, and shape and direction of the MR jet [11].
In humans, measuring the area of the regurgitant orifice during systole, i.e. planimetry of the anatomic regurgitant orifice area (AROA), using realtime three-dimensional transthoracic echocardiography (RT3DE), proved to be a feasible and accurate way to assess MR [12,14]. Moreover, analysis of the AROA has demonstrated several advantages compared with other conventional techniques, as it is relatively fast and non-invasive [4,12,13]. To the best of our knowledge, no attempt has been made in veterinary medicine to calculate the AROA using RT3DE in dogs affected by MMVD.
The aims of this study were to 1) determine feasibility and repeatability of the AROA measurements using real-time three-dimensional transthoracic echocardiography (RT3DE) in dogs affected by MMVD; 2) determine whether the AROA obtained using RT3DE relates to the severity of MR assessed by an echocardiographic scoring system (MRSS) [15] and to the American College of Veterinary Internal Medicine (ACVIM) clinical staging.
Materials and methods
The echocardiographic database of the Comparative Cardiovascular Research Laboratory of the Virginia-Maryland College of Veterinary Medicine was retrospectively searched for client- owned dogs referred to the cardiology service of the same institution in a period comprised between July 2013 and June 2015. Inclusion criteria were echocardiographic diagnosis of MMVD, availability of RT3DE data sets, and availability of a complete echocardiographic examination. The conventional echocardiographic examination comprised acquisition of standard M-mode, 2D, and Doppler blood flow measurements using standard planes [16], with continuous electrocardiographic monitoring using an ultrasound unit1 equipped with 1.5—10 MHz phased array transducers. In addition, RT3DE data sets of the mitral valve (MV) were acquired from a left apical acoustic window using the same ultrasound unit1 equipped with a matrix array transducer.2 All the RT3DE data sets were acquired using a full-volume four beats acquisition. Myxomatous mitral valve disease in included dogs was diagnosed based on the echocardiographic identification of MV thickening and/or prolapse in combination with the presence of MR identified by color Doppler. Exclusion criteria were the use of sedative drugs during echocardiographic examination and the presence of other acquired or congenital cardiovascular disorders other than MMVD.
In all dogs, the AROA was measured using commercial dedicated software.3 Three-dimensional data sets optimized for acquisition of the MV were sorted, and a data set of appropriate quality and free from stitching artifacts crossing the MV was selected for measuring the AROA. Two orthogonal long-axis planes and a third plane, orthogonal to the other two, and therefore representing a short-axis plane, were simultaneously visualized. A mid systolic frame was selected for the analysis. When the number of systolic frame was even, the operator arbitrarily chose one of the two central frames of the sequence. The long-axis planes were aligned perpendicular to the MV leaflets, to identify the assumed location of the regurgitant orifice. From this view, the short-axis plane was aligned so that it crosses the center of the regurgitant orifice. In this latter plane, the area (in cm2) of the orifice in the MV (AROA) was manually tracked [14] (Fig. 1). One observer conducted the measurements in all dogs. The number of data sets in which it was possible to measure the AROA was recorded, and the feasibility of this technique was expressed as the percentage of data sets in which the AROA could be obtained over the total dogs fulfilling the inclusion criteria. In addition, nine volume loops from nine different patients were randomly selected among the ones in which the AROA was obtained; and on these loops, the AROA measurement was performed twice by the same observer and also twice by a second observer. These data were used to assess intra-observer repeatability and inter-observer reproducibility of the AROA.
1 Artida, Toshiba Medical Systems, Tokyo, Japan.
2 PST-25SX matrix-array transducer, Toshiba Medical Systems, Tokyo, Japan.
3 4D Cardio-View 3.0 TomTec Imaging Systems, Unters- chleisssheim, Germany.
All patients were classified according to the ACVIM staging guidelines [3]. Cardiac remodeling was defined as the presence of echocardiographic evidence of left atrial (LA) enlargement (left atrial volume calculated using the biplane area-length method [17] indexed to body weight LA Vol/BW >1.1 mL/kg and/or left atrial to aortic root ratio LA:Ao >1.5) [15], and increased left ventricular end-diastolic dimensions (compared with expected dimensions) [18]. For each patient, an MRSS was also calculated using a previously reported method [15] integrated with the LA dimensions, to assess overall MR severity. The MRSS of this study was therefore determined using the following parameters: color Doppler regurgitant jet area [19], peak velocity of the mitral inflow [20], left ventricular internal dimension in diastole [21], leaflet anatomy [22], density of the continuous wave Doppler MR signal [13], and LA enlargement using LA volume (LA Vol/BW) and/or the LA:Ao, prioritizing LA Vol/BW (Table 1) [15]. Individual scores for each of the parameters were summed to obtain the MRSS, and total score assigned patients to their MR severity class (mild: 4—7, moderate: 8—12, and severe: >12). Because of our inclusion criteria, dogs in this study could have an MRSS ranging from 6 to 17.
Statistical analysis
Intra- and inter-observer repeatability of the AROA measurements was calculated as % coefficient of variation (%CV). The %CV was calculated for each dog, and is presented as mean (min—max). Furthermore, a variance component analysis was performed using a nested random effect model. The effects of dog, observer (interobserver), and measurement instance (i.e. repeated measure on the same image by the same observer) were evaluated and are reported as % of total variance. The distributions of age, body weight (BW), and AROA were tested using the Shapiro—Wilk test. Normally distributed data are presented as mean (±SD), whereas non-normally distributed data are presented as median (25th—75th percentile). Baseline characteristics of dogs in which measuring the AROA was possible or not were compared using a chi-squared test, an unpaired Student t-test, or a Wilcoxon signed rank test, as appropriate for data characteristics and distribution.
Figure 1 Obtaining the AROA from real-time three-dimensional transthoracic echocardiographic data set
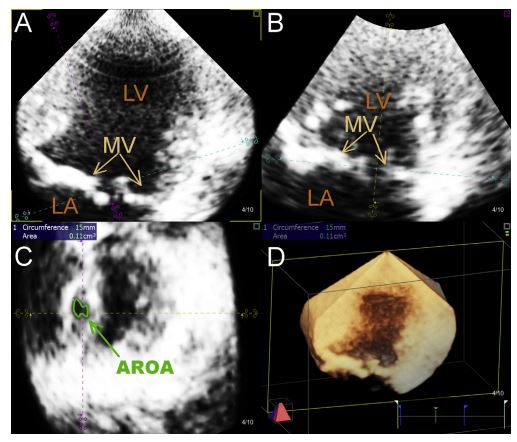
In a midsystolic frame, two orthogonal long-axis planes are moved using the pink and the yellow dotted lines parallel to the supposed regurgitation orifice and perpendicular to the MV leaflets (A and B). The short-axis plane (light blue line) is aligned so that it crosses the center of the regurgitant orifice so that planimetry of the regurgitant orifice area can be performed in the short-axis surgical view of the mitral valve obtained (C). The lower right image shows a threedimensional (3D) overview (D). LV, left ventricle; MV, mitral valve leaflets; LA, left atrium; AROA, anatomic regurgitant orifice area.
Differences in the AROA between dogs in different MRSS classes and in different ACVIM clinical stages were evaluated using a Kruskal—Wallis test. When this test disclosed statistically significant differences, post-hoc evaluation with a Dunn test was performed to determine differences between individual groups. A level of p<0.05 was considered significant for all the statistical tests.
Results
A total of 81 dogs were enrolled in the study; but in 21 patients, the AROA could not be obtained because of poor data set quality or presence of stitching artifacts in the RT3DE examinations. The feasibility of the technique was therefore 74%.
The remaining 60 dogs in which the AROA was measurable consisted of 32 males and 28 females, with a mean age of 10.25 ± 2.63 years and median BW of 9.6 kg (range 7.23—13.08 kg). Twenty-five breeds were represented. Eleven dogs were of mixed-breed origin. The most common breed was Cavalier King Charles spaniel (n = 11), followed by Shih Tzu (n = 4), Beagle (n = 3), Dachshund (n = 3), and Lhasa Apso (n = 3). The remaining 19 breeds were represented by two or less dogs each. Twenty-two dogs were classified as ACVIM stage B1, 29 as stage B2, and nine were classified as stage C. Of the 60 patients, 15 dogs were classified as mild MRSS, 28 dogs as moderate MRSS, and 17 as severe MRSS.
Dogs in which it was feasible to measure the AROA had a lower BW than dogs in which the AROA was not measurable (p=0.03). Between dogs with or without planimetry of the AROA, there was no difference in sex proportions (p=0.28) or age (p=0.27).
The inter- and intra-observer coefficients of variation of measuring the AROA were 26% (0—64%) and 21% (0—35%), respectively. Variance component analysis showed that most of the variation was due to observer (41.1%), followed by measurement instance (31.6%) and dog (27.3%).
There was no significant difference between the AROA of dogs in different ACVIM stages (p=0.170; Fig. 2). A statistically significant difference was found between the AROA and different MRSS classes (p=0.046). The AROA was significantly greater (p=0.045) in dogs with a severe MRSS (0.06 cm2 [range 0.04—0.08 cm2]) compared with the ones with mild MRSS (0.03 cm2 [range 0.02—0.06 cm2]). There was no significant difference in the AROA between dogs in mild and moderate (0.04 cm2 [range 0.03—0.06 cm2]) MRSS class (p=0.84) or moderate and severe MRSS (p=0.28; Fig. 3).
Figure 2 Box and whisker plot of the AROA in different ACVIM clinical stages
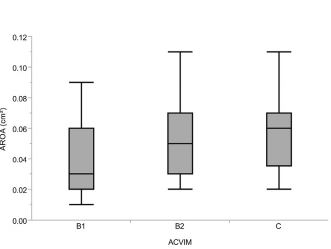
ACVIM, American College of Veterinary Internal Medicine; AROA, anatomic regurgitant orifice area.
Table 1 Classification system for MRSS for dogs with MMVD, based on echocardiographic parameters
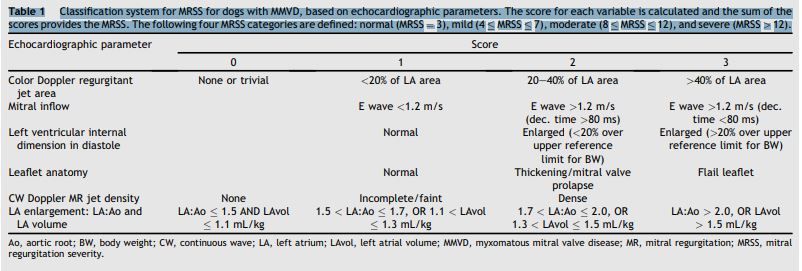
The score for each variable is calculated and the sum of the scores provides the MRSS. The following four MRSS categories are defined: normal (MRSS ¼ 3), mild (4 MRSS 7), moderate (8 MRSS 12), and severe (MRSS> 12).
Figure 3 Box and whisker plot of the AROA in different mitral regurgitation severity score classes
The asterisk indicates a significant difference. AROA, anatomic regurgitant orifice area.
Discussion
This is the first study aimed to measure the AROA in dogs affected by MMVD using RT3DE. In 74% of the RT3DE data sets, it was feasible to measure the AROA. In the other 26% (21 patients) poor image resolution or stitching artifacts did not allow measuring the AROA. The feasibility of measuring the AROA reported in human studies is generally higher [4,14,23].
Particularly, Lange et al. [4] found a feasibility of 95% when measured from the atrial side and 89% when measured from the ventricle. This higher feasibility is likely due to the fact that those studies were performed using transesophageal echocardiography, which increases resolution and therefore the quality of the data sets, compared with RT3DE. Dogs in which it was feasible to measure AROA had a significantly lower BW compared with the ones in which AROA was not measurable and this could hypothetically be due to the fact that the same matrix-array transducer was used in all patients, which might provide a better resolution and less stitching artifacts in smaller patients. This finding is in accordance with a previous report on real-time three-dimensional transthoracic echocardiography (RT3DE) in dogs using the same equipment, where the technique proved to be more feasible in smaller dogs, when used for analyzing the MV of a mixed canine population.4
The inter- and intra-observer %CVs for the AROA and the variance component analysis suggest that the observer contributed the most to the variability. Individual observer preferences on the level of zooming, brightness and contrast of the images, and the accuracy in tracing the AROA could have contributed to this. Standardizing the technique of measuring the AROA and providing more training to observers should be considered to improve the variability. However, part of the variability could be intrinsic of the technique and equipment used. In fact, the relatively low volume rate, when compared with the heart rate of dogs undergoing an echocardiographic examination, allows the acquisition of only few systolic frames. The AROA was measured in what appeared as a mid-systolic frame but, given the low temporal resolution, it is possible that the AROA was measured in different phases of the systole. Because the regurgitant orifice is dynamic during the systole [24,25], this could cause variability in the AROA measurement in the same dog, or also among our population of dogs. Also, we did not attempt to evaluate intra-patient beat-to-beat variability of AROA, which could introduce another source of variation. More, individual patients do vary in BW, thoracic conformation, fat percentage, and cooperation during ultrasound, which causes variation in resolution.
In this study, patients were classified by both ACVIM staging and MRSS. Although there is little conclusive evidence that MR severity can be used as a prognostic factor in veterinary medicine as it is in human medicine, there are various studies that support this hypothesis [26—28]. Currently, no gold standard exists for quantification of MR severity in veterinary medicine. Therefore, a combined assessment with 2DE and Doppler echocardiography is still the method of choice for noninvasive evaluation of MR severity [13,29]. The scoring system used in this study is attempting this by combining several established echocardio- graphic parameters which are either associated with a worse prognosis in both pre-clinical and clinical dogs with MMVD [9,26,30], or are validated to assess MR severity [13,19,20]. The system has been used in veterinary medicine before [15], and it is based on previously published recommendations for echocardiographic evaluation of the MR severity in humans and dogs [13,30,31]. Combining the parameters allows internal verification of interpretation and takes physiologic conditions that could alter the accuracy of certain parameters into account. Ultimately, the MRSS gives the opportunity to compare MR severity with the AROA.
4 Menciotti G, Borgarelli M., Wesselowski S., Abbott J. Quantitative evaluation of canine mitral valve in dogs using threedimensional echocardiography. In: Research Communications of the 24th ECVIM-CA Congress. J Vet Intern Med 2015;29:423- 484.
The AROA did not show any difference between ACVIM stages. This is not an unexpected result. In fact, this classification is meant for linking appropriate treatment recommendations to the clinical stages, and it does not strictly evaluate MR severity. Although partially related to the development of clinical sign, MR severity is not the only determinant for the development of clinical signs. Hence, it was not expected that the AROA would have been related to ACVIM staging, whereas the difference found between the AROA and MRSS classes mild and severe was expected, as it can be hypothesized that a bigger AROA would result in a more severe MR, and therefore more severe MRSS.
Most limitations of this study are related to its retrospective design. Some dogs were excluded because they had incomplete echocardiographic studies or real-time three-dimensional transthoracic echocardiography (RT3DE) data sets where of insufficient quality. Also, the AROA was not compared with other echocardiographic methods for evaluating MR, like the effective regurgitant orifice area (EROA) or the vena contracta width. In human medicine, there is a good correlation between AROA and EROA; and in one study, the former was found to be more reproducible than the latter, although the bias between the two techniques increases in patients with eccentric jets [4,27]. However, it is necessary to underline that in the referenced studies, AROA was calculated using three-dimensional transesophageal echocardiography, which has a better spatial resolution compared with RT3DE, but would have limited application in clinical veterinary settings because of the need of general anesthesia to be performed. Also, the conventional estimation of EROA presents some limitations related to the common geometry of MR jets in dogs (i.e. eccentric, and with asymmetric vena contracta). This could be overcome by real-time three-dimensional transthoracic echocardiography (RT3DE) techniques, which allow a more complex analysis of regurgitant jets [32]. However, our data sets did not comprise RT3DE color flow Doppler data, and therefore a comparison between AROA and RT3DE-derived EROA was not possible in the present study.
Another limitation is that we cannot rule out unconscious bias because of the lack of a blind study design. Although ACVIM stage and MRSS score could have been blinded, it is not possible to avoid expectations on disease severity based on subjective echocardiographic findings while measuring the AROA. Lastly, there is a limited number of dogs enrolled in the study and an imbalance in patient groups. In fact, only nine dogs were in stage C, against 22 and 29 in B1 and B2, respectively. This might have limited our ability to detect differences between these groups. According to MRSS classes instead, dogs are more equally distributed, but each individual class comprises relatively few dogs.
Conclusions
This study shows that it is feasible to obtain the AROA in dogs with MMVD using real-time three-dimensional transthoracic echocardiography (RT3DE). The AROA of patients with severe MR, as assessed by a combination of conventional echocardiographic variables, is greater than the one of patients with mild MR. Further standardization of the technique is required to improve inter- and intra-observer variability, and investigate the diagnostic and prognostic potential of this variable.
Conflicts of Interest Statement
The authors do not have any conflicts of interest to disclose.
Acknowledgments
Sandra Muller performed this study at the Department of Small Animal Clinical Sciences, Virginia-Maryland College of Veterinary Medicine, Blacksburg, VA as part of the Merial Veterinary Scholars Program 2015. The authors acknowledge the help of Dr. Mark Dirven from Utrecht University in the manuscript preparation.
References
- Detweiler DK, Patterson DF. The prevalence and types of cardiovascular disease in dogs. Ann N Y Acad Sci 1965;127: 481-516.
- Darke P. Valvular incompetence in Cavalier King Charles Spaniels. Vet Rec 1987;120:365-6.
- Atkins C, Bonagura J, Ettinger S, Fox P, Gordon S, Haggstrom J, Hamlin R, Keene B, Luis-Fuentes V, Stepien R. Guidelines for the diagnosis and treatment of canine chronic valvular heart disease. J Vet Intern Med 2009;23:1142-50.
- Lange A, Palka P, Donnelly JE, Burstow DJ. Quantification of mitral regurgitation orifice area by 3-dimensional echocardiography: comparison with effective regurgitant orifice area by PISA method and proximal regurgitant jet diameter. Int J Cardiol 2002;86:87-98.
- Grigioni F, Enriquez-SaranoM, ZehrKJ, Bailey KR, Tajik AJ. Ischemic mitral regurgitation: long-term outcome and prognostic implications with quantitative Doppler assessment. Circulation 2001;103:1759-64.
- Thavendiranathan P, Phelan D, Collier P, Thomas JD, Flamm SD, Marwick TH. Quantitative assessment of mitral regurgitation: how best to do it. JACC Cardiovasc Imaging 2012;5:1161-75.
- Enriquez-Sarano M, Avierinos J-F, Messika-Zeitoun D, Detaint D, Capps M, Nkomo V, Scott C, Schaff HV, Tajik AJ. Quantitative determinants of the outcome of asymptomatic mitral regurgitation. N Engl J Med 2005; 352:875-83.
- Wesselowski S, Borgarelli M, Menciotti G, Abbott J. Echo- cardiographic anatomy of the mitral valve in healthy dogs and dogs with myxomatous mitral valve disease. J Vet Cardiol 2015;17:97-106.
- Sargent J, Muzzi R, Mukherjee R, Somarathne S, Schranz K, Stephenson H, Connolly D, Brodbelt D, Fuentes VL. Echo- cardiographic predictors of survival in dogs with myxomatous mitral valve disease. J Vet Cardiol 2015;17:1-12.
- Ljungvall I, Hoglund K, Carnabuci C, Tidholm A, Haggstrom J. Assessment of global and regional left ventricular volume and shape by real-time 3-dimensional echocardiography in dogs with myxomatous mitral valve disease. J Vet Intern Med 2011;25:1036-43.
- Di Marcello M, Terzo E, Locatelli C, Palermo V, Sala E, Dall'Aglio E, Bussadori CM, Spalla I, Brambilla PG. Assessment of mitral regurgitation severity by Doppler color flow mapping of the vena contracta in dogs. J Vet Intern Med 2014;28:1206-13.
- Hamada S, Altiok E, Frick M, Almalla M, Becker M, Marx N, Hoffmann R. Comparison of accuracy of mitral valve regurgitation volume determined by three-dimensional transesophageal echocardiography versus cardiac magnetic resonance imaging. Am J Cardiol 2012;110: 1015-20.
- Zoghbi WA, Enriquez-Sarano M, Foster E, Grayburn PA, Kraft CD, Levine RA, Nihoyannopoulos P, Otto CM, Quinones MA, Rakowski H, Stewart WJ, Waggoner A, Weissman NJ, Grayburn MD, William J. Recommendations for evaluation of the severity of native valvular regurgitation with two-dimensional and Doppler echocardiography. J Am Soc Echocardiogr 2003;16:777-802.
- Altiok E, Hamada S, van Hall S, Hanenberg M, Dohmen G, Almalla M, Grabskaya E, Becker M, Marx N, Hoffmann R. Comparison of direct planimetry of mitral valve regurgitation orifice area by three-dimensional transesophageal echocardiography to effective regurgitant orifice area obtained by proximal flow convergence method and vena contracta area determined by color. Am J Cardiol 2011; 107:452-8.
- Wesselowski S, Borgarelli M, Bello NM, Abbott J. Discrepancies in identification of left atrial enlargement using left atrial volume versus left atrial-to-aortic root ratio in dogs. J Vet Intern Med 2014;28:1527-33.
- Thomas WP, Gaber CE, Jacobs GJ, Kaplan PM, Lombard CW, Moise NS, Moses BL, Vet M. Recommendations for Standards in transthoracic twodimensional echocardiography in the dog and cat. J Vet Intern Med 1993;7:247-52.
- Lang RM, BierigM, Devereux RB, Flachskampf FA, Foster E, Pellikka PA, Picard MH, Roman MJ, Seward J, Shanewise JS, Solomon SD, Spencer KT, Sutton MSJ, Stewart WJ. Recommendations for chamber quantification: a report from the American Society of Echocardiography's Guidelines and Standards Committee and the Chamber Quantification Writing Group, developed in conjunction with the European Association of Echocardiograph. J Am Soc Echo- cardiogr 2005;18:1440-63.
- Cornell CC, Kittleson MD, Torre P Della, Haggstrom J, Lombard CW, Pedersen HD, Vollmar A, Wey A. Allometric scaling of M-mode Cardiac measurements in normal adult dogs. J Vet Intern Med 2004;18:311-21.
- Muzzi RAL, de Araujo RB, Muzzi LAL, Pena JLB, Silva EF. Regurgitant jet area by Doppler color flow mapping: quantitative assessment of mitral regurgitation severity in dogs. J Vet Cardiol 2003;5:33-8.
- Kim J-H, Park H-M. Usefulness of conventional and tissue Doppler echocardiography to predict Congestive heart failure in dogs with myxomatous mitral valve disease. J Vet Intern Med 2015;29:132-40.
- Stewart JA, Wei CC, Brower GL, Rynders PE, Hankes GH, Dillon AR, Lucchesi PA, Janicki JS, Dell'Italia LJ. Cardiac mast cell- and chymase-mediated matrix metalloproteinase activity and left ventricular remodeling in mitral regurgitation in the dog. J Mol Cell Cardiol 2003;35:311-9.
- Terzo E, Di Marcello M, McAllister H, Glazier B, Lo Coco D, Locatelli C, Palermo V, Brambilla PG. Echocardiographic assessment of 537 dogs with mitral valve prolapse and leaflet involvement. Vet Radiol Ultrasound 2009;50: 416-22.
- Breburda CS, Griffin BP, Pu M, Rodriguez L, Cosgrove DM, Thomas JD. Three-dimensional echocardiographic planimetry of maximal regurgitant orifice area in myxomatous mitral regurgitation: intraoperative comparison with proximal flow convergence. J Am Coll Cardiol 1998;32: 432-7.
- Buck T, Plicht B, Kahlert P, Schenk IM, Hunold P, Erbel R. Effect of dynamic flow rate and orifice area on mitral regurgitant stroke volume quantification using the proximal isovelocity surface area method. J Am Coll Cardiol 2008;52:767-78.
- Paiva RMP, Garcia-Guasch L, Manubens J, Montoya- Alonso JA. Proximal isovelocity surface area variability during systole in dogs with mitral valve prolapse. J Vet Cardiol 2011;13:267-70.
- Borgarelli M, Savarino P, Crosara S, Santilli RA, Chiavegato D, Poggi M, Bellino C, La Rosa G, Zanatta R, Haggstrom J, Tarducci A, La Rosa G. Survival characteristics and prognostic variables of dogs with mitral regurgitation attributable to myxomatous valve disease. J Vet Intern Med 2008;22:120-8.
- Chandra S, Salgo IS, Sugeng L, Weinert L, Settlemier SH, Mor-Avi V, Lang RM. A three-dimensional insight into the complexity of flow convergence in mitral regurgitation: adjunctive benefit of anatomic regurgitant orifice area. Am J Physiol Heart Circ Physiol 2011;301:H1015-24.
- Olsen LH, Mow T, Koch J, Pedersen HD. Heart rate variability in young, clinically healthy Dachshunds: influence of sex, mitral valve prolapse status, sampling period and time of day. J Vet Cardiol 1999;1:7-16.
- Gouni V, Serres FJ, Pouchelon J-L, Tissier R, Lefebvre HP, Nicolle AP, Sampedrano CC, Chetboul V. Quantification of mitral valve regurgitation in dogs with degenerative mitral valve disease by use of the proximal isovelocity surface area method. J Am Vet Med Assoc 2007;231: 399-406.
- Borgarelli M, Crosara S, Lamb K, Savarino P, La Rosa G, Tarducci A, Haggstrom J, Santilli RA, Chiavegato D, Poggi M, Bellino C, La Rosa G, Zanatta R, Haggstrom J. Survival characteristics and prognostic variables of dogs with preclinical chronic degenerative mitral valve disease attributable to myxomatous degeneration. J Vet Intern Med 2012;26:69-75.
- Haggstrom J, Boswood A, O'Grady M, Jons O, Smith S, Swift S, Borgarelli M, Gavaghan B, DiFruscia R, et al. Effect of pimobendan or benazepril hydrochloride on survival times in dogs with congestive heart failure caused by naturally occurring myxomatous mitral valve disease: the QUEST study. J Vet Intern Med 2008;22:1124-35.
- Tidholm A, Bodegard-Westling A, Hoglund K, Haggstrom J, Ljungvall I. Real-time 3-dimensional echocardiographic assessment of effective regurgitant orifice area in dogs with myxomatous mitral valve disease. J Vet Intern Med 2017;31:303-10.
^Наверх









