Analysis of weight uniformity, content uniformity and 30-day stability in halves and quarters of routinely prescribed cardiovascular medications

Author information
Margiocco M.L., Warren J., Borgarelli M., Kukanich B. Analysis of weight uniformity, content uniformity and 30-day stability in halves and quarters of routinely prescribed cardiovascular medications // J Vet Cardiol. 2009 Jun;11(1):31-9.
Abstract
OBJECTIVES: Congenital and acquired cardiac disorders are frequently diagnosed in small breed dogs and cats. In order to appropriately dose cardiovascular drugs for small patients, fractions of commercially available tablets must be prescribed. The aims of this study were to evaluate weight and content uniformity and 30-day chemical stability in halves and quarters of 11 drug formulations commonly prescribed to treat cardiovascular disorders in small breed dogs and cats.
ANIMALS, MATERIALS AND METHODS: Fifteen tablets from 11 drug formulations were obtained within the same lot. Tablets were split by a single operator using a commercially available pill splitter. Whole tablets, halves and quarters were weighed and stored in plastic containers. High-pressure liquid chromatography or liquid chromatography with mass spectrometry were utilized to determine drug content and repeated 30 days later to estimate chemical stability.
RESULTS: Statistically significant weight variability, content non-uniformity and chemical degradation were found for some formulations. Digoxin showed a significant degradation that should be considered in clinical practice.
CONCLUSIONS: It appears that pill splitting is overall a relatively reliable practice; however tablets should not be split ahead of time but only immediately prior to intended usage.
Introduction
Cardiovascular disease is a well-recognized cause for morbidity and mortality in both dogs and cats. It has been estimated to affect approximately 11% of dogs and 10.4% of cats evaluated by veterinarians or at necropsy, respectively.1,2 Chronic degenerative valve disease (CDVD) is the most common acquired cardiac disorder in dogs, and most frequently affects canine patients less than 15 kg.3,4 Due to the size of these patients (small breed dogs and cats), available tablet formulations and their respective therapeutic dosages (Table 1), many common cardiac drugs must be split into halves or quarters.6-15 There is little information to provide evidence for or against pill splitting. Besides the need for tablet splitting in patients with a low body weight, when a dosage form of the required strength is not commercially available, other potential reasons exist. Examples are a flexible dosing regimen (i.e. adapting the dose of furosemide to the specific needs of the patient), slow up-titration of a drug (i.e. to reduce undesired side-effects), or slow down-titration (i.e. to identify the lowest effective dose or to wean a patient off a medication).
There are no established criteria for evaluation of dosage uniformity in tablet fractions obtained by pharmacists or patients.15 Most studies on split tablets adopted, assuming uniform content, the United States Pharmacopeia (USP) Standard, that allows for a 15% deviation in weight from the label claim and for a Relative Standard Deviation (RSD, i.e. the absolute value of the coefficient of variability, CV) of not greater than 6%.16,17 Although these are the only available guidelines, they have been developed for intact dosage units and may not be appropriate for the analysis of data from fractionated tablets. Three studies looked at the clinical reliability of chronic treatment based on split tablets involving lisinopril18 and statins.19,20 No differences in clinical parameters (systemic blood pressure and lipid profile, respectively) were found when comparing equal strength full tablet vs. split tablet treatment. On the basis of these data, pharmacokinetic (PK) and pharmacodynamic (PD) considerations, the use of a split tablet is generally accepted in chronic therapy with drugs characterized by a long half-life and wide therapeutic margins.21,22 Several studies looked at the weight uniformity of fractions of different pharmaceutical products and found significant deviations (range 10% to greater than 20%) from the ideal weight, with only 31.8% of the 22 prescriptions evaluated in one study meeting the USP guidelines reported.15,23,24 Whether this degree of variability carries a clinical risk mainly depends on the therapeutic window and dose—response curve for each specific drug. However these studies show the existence of potentially unacceptable variability in the weight of tablet fractions.
It must be emphasized that all of these studies assumed homogeneous distribution of the active principle within the whole tablet. While relatively strict guidelines exist that regulate the degree of content uniformity in scored tablets, this does not necessarily apply to the production of unscored tablets.
Table 1 Commercially available strength, average dose, and range of body weight (BW) compatible with a full- tablet dosing regimen (rounded to the closest whole number)
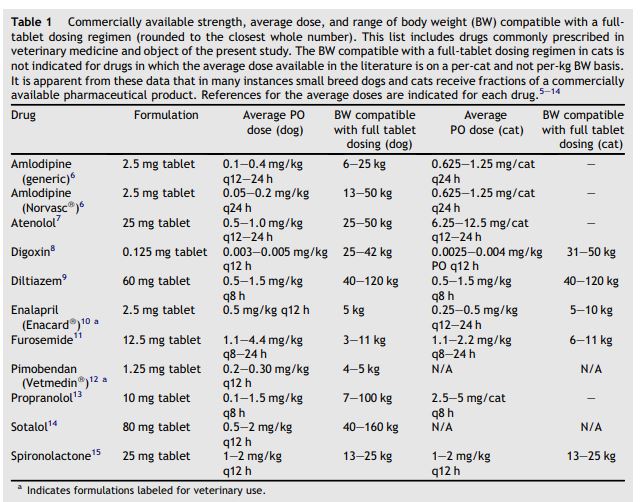
This list includes drugs commonly prescribed in veterinary medicine and object of the present study. The BW compatible with a full-tablet dosing regimen in cats is not indicated for drugs in which the average dose available in the literature is on a per-cat and not per-kg BW basis. It is apparent from these data that in many instances small breed dogs and cats receive fractions of a commercially available pharmaceutical product. References for the average doses are indicated for each drug.5-14
a Indicates formulations labeled for veterinary use.
Splitting unscored tablets is currently considered "off-label’’ because of the potential lack in content uniformity. One study showed the existence of significant content dishomogeneity in the simvastatin tablets produced by secondary manufacturers, raising the additional concern about the potential for a greater degree of variability in generic formulations compared to brand name products.25 Another important factor to be considered is that the active principle may not be stable after splitting; this may be particularly true for coated formulations, but theoretically holds true for any drug, since splitting increases the surface area in direct contact with air, humidity, and potentially, light.26 An extreme example is the well known instability of soluble aspirin that limits the use of a split tablet to only 24 h.27
Therefore, several concerns exist when splitting pills for administration. The purpose of this study was to specifically investigate some of these factors in 10 commonly prescribed cardiac drugs (11 formulations). In particular we aimed at testing the following hypotheses: 1) pill splitting leads to even halves and quarters (weight uniformity), 2) the active principle is evenly distributed within each commercially available tablet, even in products not manufactured as scored tablets (content uniformity), 3) scored tablets are characterized by a more uniform distribution of their content compared to unscored ones, and 4) the active principle contained in quartered tablets remains stable when stored in sealed plastic containers, protected from light, for 30—45 days (chemical stability).
Animals, materials and methods
Fifteen tablets from each of the following 11 drug formulations were obtained within the same lot: amlodipine (generic) 2.5 mg,c amlodipine (Nor- vasc®) 2.5 mg,d atenolol (generic) 25 mg,e digoxin (generic) 0.125 mg,f diltiazem (generic) 60 mgg (only 10 tablets were available), enalapril (Enacard®) 2.5 mg,h furosemide (generic) 12.5 mg,* i pimobendan (Vetmedin®) 1.25 mg,j propranolol (generic) 10 mg,k sotalol (generic) 80 mg,l and spironolactone (generic) 25 mg.m Ten of the whole tablets of each drug were individually weighed using an electronic balancen and the mass of each tablet was recorded to the one-tenth of a milligram (0.1 mg). The calibration of the balance was confirmed prior to the start of the study and at the conclusion of the study. Using a standard guillotine pill splittero and a single operator, tablets were then split into halves. Between each cut, the pill splitter was rinsed with methanol, wiped with a cleaning tissuep and allowed to dry. Each half was individually weighed in a clean, disposable weigh boat. The procedure was repeated for quarters. Quarter tablets were placed into labeled 0.5 g plastic vials, and sealed. The shape of each pharmaceutical product was recorded as well as a subjective comment from the operator about difficulties encountered with each specific drug during the procedure of pill splitting (Table 2).
High-pressure liquid chromatography (HPLC) analysis with ultraviolet absorption was used to determine the drug content within each tablet or quarter, except for enalapril in which liquid chromatography with mass spectrometry (LC/MS) was used to determine drug content. Standard curves were constructed daily using authenticated reference standards, except for pimobendan in which an authentic reference standard was not available. Standard curves were accepted if they were linear, the coefficient of determination (r2) was at least 0.99, and the predicted values were within 15% of the actual values. The standard curve for pimobendan was constructed using Vetmedin® (pimobendan) 1.25 mg tablets initially dissolved in 90% methanol and 10% 0.1% tri- fluoroacetic acid and subsequently diluted in 50% methanol. Confirmation of pimobendan was accomplished using LC/MS with an m/z of 335. Stability of the standards for all drugs was confirmed by reinjecting the standards at the end of each run and was acceptable if the predicted values were within 15% of the actual values.
c Mylan, Canonsburg, PA, USA. d Pfizer Labs, NY, NY, USA. e Sandoz, Princeton, NJ, USA. f Caraco, Detroit, MI, USA. g Teva, North Wales, PA, USA. h Merial, LTD, Duluth, GA, USA. i Intervet, Millsboro, DE, USA. j Boehringer Ingelheim, St. Joseph, MO, USA. k Pliva/Barr Labs, Montvale, NJ, USA.l Sandoz, Princeton, NJ, USA. m Sandoz, Princeton, NJ, USA. n Acculab AL-64, Edgewood, NY, USA. o EZY Dose, Apothecary Products, Inc, Minneapolis, MN, USA. p KimWipe, Kimberly—Clark Corporation, Roswell, GA, USA.
Table 2 The shape of the tablets, whether the products are manufactured as a scored (Y) or unscored (N) tablet, and a subjective evaluation on the action of splitting each drug are reported
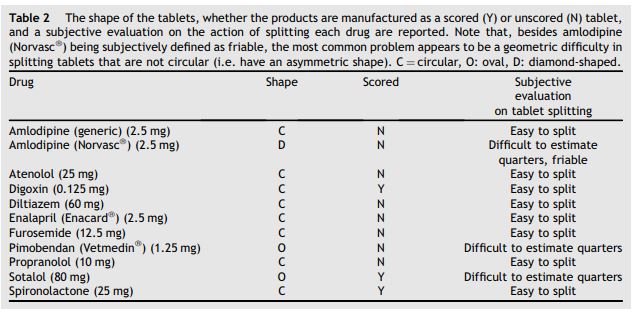
Note that, besides amlodipine (Norvasc®) being subjectively defined as friable, the most common problem appears to be a geometric difficulty in splitting tablets that are not circular (i.e. have an asymmetric shape). C = circular, O: oval, D: diamond-shaped.
The recovery of drug from whole tablets was assessed using HPLC in order to assure high drug recovery for all tablets, except diltiazem due to the limited number of tablets available within the available brand and lot. Quartered tablets, 2 per tablet, were randomly selected for high-pressure liquid chromatography analysis using a random number generator on a portable calculatorq so the quarters were selected from different halves of the tablet and from 10 different tablets (a total of 20 replicates per drug). Baseline analysis was performed within 7 days of tablet splitting for all drugs except pimobendan and enalapril, which were only assessed on day 30 in an identical manner. The 30-day stability was assessed on all drugs (at least 30 days after quartering, but not longer than 45 days after quartering), except for enalapril and pimobendan (previously described) by randomly assaying 1 quartered tab section per tablet from 10 different tablets (a total of 10 replicates per drug).
Statistical analyses for the weight distribution of the tablets were performed using commercially available statistical software/ Normality tests were run on all data sets (Kolmogorov—Smirnov test (KS test) with Dallal—Wilkinson—Lilliefor p value, a D’Agostino & Pearson omnibus normality test, and a Shapiro—Wilk normality test). A paired Student’s t test was used to compare the means of the distribution of weights between the two halves obtained from the same whole tablet and between the two quarters obtained from the same half. Similar comparisons were conducted between the measured weight and the expected weight of halves and quarters, where the expected weights were calculated dividing the measured weight of the whole tablet by 2 and 4, respectively. Analysis of Variance (ANOVA) was also used to compare the means of the weight distribution between the four quarters obtained from the same whole tablet. Statistical significance was set as p < 0.05. CV% values were rounded to the closest whole percentage.
q fx-991h, Casio, Inc, Dover, NJ, USA.
Statistical analysis of drug content by HPLC and LC/MS was performed using commercially available statistical software/ Normality tests (KS test) were run on drug content within the quartered tablets and indicated that the distribution was not normal for all the drugs, therefore descriptive statistics included the mean and 5th and 95th percentiles. To assess the uniformity of the drug within the tablet, the correlation of the weight of the tablet quarters and drug content of the tablet quarters was determined by the Spearman Rank Order Correlation test, due to the data not being normally distributed (n = 20 for each drug), and the correlation reported as the correlation coefficient. The drug content for the quartered tablets from each half tablet was compared for differences after initial splitting using the Mann—Whit- ney Rank Sum test and the 30-day content was compared to the initial content also using the Mann—Whitney Rank Sum test. Percentage content values were rounded to the closest whole percentage.
r Prism 4 for Macintosh, GraphPad Software, Inc., La Jolla, CA, USA.s SigmaStat 3.11, Systat Software, Inc., San Jose, CA, USA.
Results
Table 3 shows the weight of the whole tablets, halves and quarters. Data on weight (mass) were normally distributed and are reported as mean, SD, and CV of the mean. There was a statistically significant difference in the mean weight of the two halves obtained from the same whole tablet for atenolol 25 mg and Vetmedin® (pimobendan) 1.25 mg (p = 0.012 and p = 0.014, respectively, from a paired Student’s t test), while there were no significant differences in the other tablets tested. There was a statistically significant difference in the mean weight of the two quarters obtained from the same half of a tablet for diltiazem 60 mg (p = 0.044 from a paired Student’s t test, result also confirmed by an ANOVA test comparing the mean weight of the four quarters from the same whole tablet, data not shown). Table 4 shows the mean difference and the 95% CI for the statistically significant results from Table 3 (i.e. atenolol and pimobendan halves and diltiazem quarters). The magnitudes of these differences are expressed as a percentage of the measured weight of each parent tablet (i.e. the whole tablet for the halves and the half tablet for the quarters). Table 4 also shows how these mean percentage differences in mass translate into an absolute content difference, assuming homogeneous distribution of the active principle within the tablets analyzed.
The CV of the weight for the halved tablets was greater than 6% in the following formulations: amlodipine (generic), atenolol, diltiazem, enalapril, furosemide, amlodipine (Norvasc®), propranolol, and spironolactone. Enalapril showed the highest CV (18%) in the halved fraction. All the formulations showed a CV of greater than 6% in the quartered fraction, with amlodipine (Norvasc®) showing the highest value (31%) (Table 3).
The results of the drug content as determined by HPLC and LC/MS are presented in Table 5. The 5th and 95th percentiles of the expected content were within ±20% in whole tablets for all tested drugs except digoxin which was 108—128% of the expected content. Only digoxin had the actual drug content (5th and 95th percentiles) within ±20% of the targeted drug concentration in the quartered tablets. Digoxin quartered tablets had significantly decreased drug content after 30 days of storage, decreasing from an initial mean content of 105% of the expected content to 68% of the expected content. Significant decreases in the expected drug content after 30 days of storage also occurred with amlodipine (generic), amlodipine (Norvasc®), and spironolactone, although the magnitude of the decrease was not as dramatic as digoxin. The correlation coefficient of the weight of the quartered tablet and the measured content in the quartered tablets was greater than 0.7 for amlodipine (Norvasc®), atenolol, digoxin, enalapril (Enacard®), furosemide, pimobendan (Vetme- din®), propranolol, and sotalol suggesting that the drug was relatively uniformly distributed in these tested tablets. A coefficient of correlation of less than 0.7 was obtained for amlodipine (generic), diltiazem, and spironolactone, suggesting that the active principle may not be homogeneously distributed within these formulations.
Table 3 Weight of whole tablets, halves and quarters are indicated as mean, SD, CV%
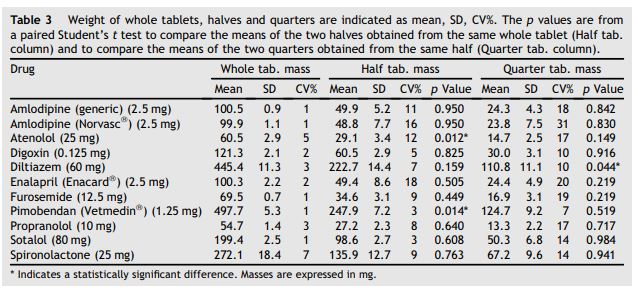
The p values are from a paired Student’s t test to compare the means of the two halves obtained from the same whole tablet (Half tab. column) and to compare the means of the two quarters obtained from the same half (Quarter tab. column).
Table 4 Data are shown for the statistically significant results from Table 3
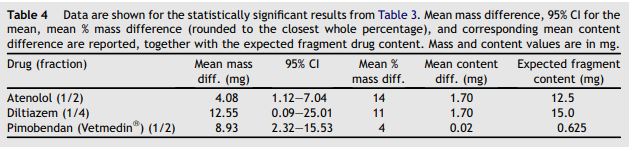
Mean mass difference, 95% CI for the mean, mean % mass difference (rounded to the closest whole percentage), and corresponding mean content difference are reported, together with the expected fragment drug content. Mass and content values are in mg.
Discussion
Cardiovascular diseases are approached in the majority of the cases with pharmacologic treatment. Data on the PK, PD and clinical characteristics of many cardiovascular drugs have been published, although are not available for all the drugs in all the veterinary species. Treatment of cardiac diseases is a chronic treatment that is virtually always maintained for a long period of
Table 5 Summary of the results of the HPLC and LC/MS determination of the relative concentration of the active principle in the 11 pharmaceutical products that were the object of the study time (often for the rest of the patient’s life), and it commonly consists of a multi-drug approach. All these aspects contribute to potential concerns regarding owner compliance and overall efficacy of long-term oral treatments. The study outlined here aimed at investigating potential pitfalls of chronic oral treatment based on split tablets, focusing on the phase preceding oral absorption. To the best of our knowledge, studies on the reliability of oral treatment based on administration of fractions of tablets have not been conducted in veterinary medicine.
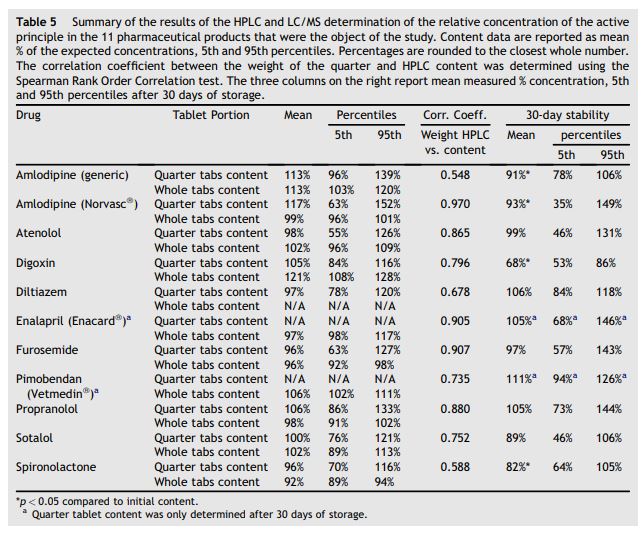
Content data are reported as mean % of the expected concentrations, 5th and 95th percentiles. Percentages are rounded to the closest whole number. The correlation coefficient between the weight of the quarter and HPLC content was determined using the Spearman Rank Order Correlation test. The three columns on the right report mean measured % concentration, 5th and 95th percentiles after 30 days of storage.
Only data for diltiazem, pimobendan (Vetme- din®) and atenolol reached statistical significance when comparing the means of the distribution of weights of halved and quartered tablets. It is important to critically evaluate the results of any statistical tool in terms not only of statistical significance (p value), but also in terms of scientific (in this case clinical) significance and overall variability (i.e. SD, or better CV). Our data showed high variability, that probably limited the number of statistically significant findings, but that, by itself is an index of poor homogeneity in the final products of pill splitting.
In our study, the weight variability of the halved fractions for many drugs did not meet the USP Standard of a CV < 6%. Some formulations showed a CV for the halves of greater than 10% (amlodipine (generic), amlodipine (Norvasc®), atenolol, and enalapril (Enacard®) — Table 3). Quartering a tablet appears to consistently result in an unacceptably high degree of weight variability, as shown by a CV of greater than 6% for all formulations, and of greater than 10% for all the formulations, except pimobendan (Vetmedin®). Amlodipine (Norvasc®) showed the highest CV (31%) for the quarters. A potential explanation for this high variability resides in the irregular shape of the product (diamond-shaped tablet) that makes it difficult to position the tablet in the loader of the pill splitter. Furthermore, amlodipine (Norvasc®) was the only formulation subjectively evaluated as "friable’’, which carries the risk for the unpredictable loss of product in the form of powder or "crumbs".
Other formulations came in a non-symmetric, oval shape (pimobendan (Vetmedin®) and sotalol, and were subjectively characterized by difficulty in estimating quarters. However, pimobendan showed a low CV for the halves and quarters, while sotalol showed a low CV for the halves and a moderate CV for the quarters. These findings may indicate that the oval shape of these specific formulations may not represent by itself a problem in the action of splitting the tablets using the pill splitter adopted in this study. It is important to recognize that different pill splitters may perform differently, as already reported in the medical literature.21
Results from the content uniformity analysis (Table 5) showed good mean values for the % content (range from 92% to 117% for both whole tablets and quarters). However, when the 5th and 95th percentiles for some quartered drugs are examined, our data indicate that the potential risk for under-dosing and/or over-dosing exists, as a result of the high variability of the expected content in the quarters. This was especially true for atenolol, amlodipine (Norvasc®), and furosemide. These data suggest poor uniformity in some commercially available products as a result of pill splitting (unequal splitting) and/or poor content uniformity. To isolate the effect of unequal distribution of the active principle within the parent tablets a correlation analysis between the measured weight of the quarters and the corresponding measured content was performed. Using the arbitrary cut-off level of 0.7 for the coefficient of variability, poor correlation was defined as an r < 0.7.< /p>
Based on our results, amlodipine (generic), diltiazem and spironolactone showed evidence of non-uniform distribution of the active principle. Amlodipine is routinely administered to cats in the form of halved or quartered tablets. The generic formulation is relatively less expensive compared to the brand name and frequently prescribed in our institution. Although the generic formulation showed evidence of content non-uniformity (r = 0.5), its round shape allowed for less weight variability as a result of pill splitting (CV for the quarters: 18%; 5th and 95th percentiles respectively 96% and 139%). Hence a trade off between shape and content uniformity has to be considered when choosing between these two commercially available formulations of amlodipine. Furthermore, these data only apply to the manufacturer and lot analyzed in this study. Scored tablets did not appear to perform differently compared to unscored tablets. A scored formulation, assumed to be split into halves, would be expected to show a better content uniformity compared to an unscored one. This hypothesis was not supported by our data. In fact of the three scored formulations analyzed (digoxin, spironolactone and sotalol), spironolactone showed one of the lowest correlation coefficients (0.6). Scored formulations may be relatively easy to split into halves, but not necessarily easy to further split into quarters.
The results of this study confirm that the 30-day chemical decay for some quartered products may be significant from a clinical standpoint. Statistically significant decreases in the active principle were documented for digoxin, both amlodipine formulations (generic and Norvasc®), and spironolactone. Digoxin showed the most dramatic reduction, with a mean 30-day measured content of only 68% of the expected value. As a consequence of its narrow therapeutic index, this magnitude of decay may be clinically significant. However, given the long pharmacokinetic half-life of digoxin, only repeated and consecutive administration of a "degraded quarter’’ would be expected to cause a clinically significant effect. The data on 30-day stability concerning spironolactone and the two amlodipine formulations, although statistically significant, are probably not clinically significant. However, prescriptions are often conserved for more than 30 days, and the potential for a more significant degradation in a longer time frame should be considered, especially for these three products.
A limitation of our study was the adoption of a single operator (a senior veterinary student) and a single type/brand pill splitter, which allowed for the control of several potential confounding variables; however our data may not apply to pills split by pet owners or the use of different pill splitters. Furthermore our findings may not be applicable to pelleted or layered medications.
Conclusions
Variability exists in the overall result of pill splitting, as documented by the present study. Sources of variability reside in either the action of pill splitting, the existence of content non-uniformity in commercially available formulations, and the risk of chemical degradation of quartered tablets. The clinical significance of this variability is difficult to quantify, and was not the goal of the study outlined here.
Some practical recommendations can be drawn from our results. In general it appears safe to split the specific formulations assessed in our research. Given the documented high variability and the potential for degradation of fractions of tablets, it seems safer to avoid splitting a large number of tablets ahead of time. Splitting one tablet at a time allows for automatic compensation in the relative content of the halves/quarters. If the morning dose contains less active principle, the evening dose will contain a proportionally greater amount of active principle. This practice will also limit the risk of degradation of the product. This appears to be a fundamental precaution for digoxin, given its significant chemical decay. However, it is important to realize that the actual bioavailability/bio- equivalence of the split tablets administered to clinical cases was not assessed which may increase or decrease the pharmacodynamic variability.
Acknowledgements
The authors would like to thank Dr. Eugene DeDonder and Dr. Shirley Arck for reviewing the paper and providing their expertise.
References
- Buchanan JW. Causes and prevalence of cardiovascular disease. In: Kirk RW, Bonagura JD, Osborne CA, editors. Current veterinary therapy XI. Philadelphia: WB Saunders; 1992. p. 647-654.
- Liu SK. Pathology of feline heart diseases. Vet Clin North Am 1997;7:323.
- Detweiler DK, Patterson DF. The prevalence and types of cardiovascular disease in dogs. Ann N Y Acad Sci 1965;127: 481-516.
- Buchanan JW. Prevalence of cardiovascular disorders. In: Fox PR, Sisson D, Moise NS, editors. Textbook of canine and feline cardiology. 2nd ed. Philadelphia: WB Saunders; 1999. p. 457-470.
- Plumb DC. Amlodipine besylate. Plumb’s veterinary drug handbook. 5th ed. Ames: Blackwell; 2005. p. 37-38.
- Plumb DC. Atenolol. Plumb’s veterinary drug handbook. 5th ed. Ames: Blackwell; 2005. p. 68-69.
- Plumb DC. Digoxin. Plumb’s veterinary drug handbook. 5th ed. Ames: Blackwell; 2005. p. 251 -254.
- Plumb DC. Diltiazem. Plumb’s veterinary drug handbook. 5th ed. Ames: Blackwell; 2005. p. 256-258.
- Plumb DC. Enalapril. Plumb’s veterinary drug handbook. 5th ed. Ames: Blackwell; 2005. p. 292-294.
- Plumb DC. Furosemide. Plumb’s veterinary drug handbook. 5th ed. Ames: Blackwell; 2005. p. 354-357.
- Plumb DC. Pimobendan. Plumb’s veterinary drug handbook. 5th ed. Ames: Blackwell; 2005. p. 637-638.
- Plumb DC. Propranolol. Plumb’s veterinary drug handbook. 5th ed. Ames: Blackwell; 2005. p. 672-674.
- Plumb DC. Sotalol. Plumb’s veterinary drug handbook. 5th ed. Ames: Blackwell; 2005. p. 710-711.
- Plumb DC. Spironolactone. Plumb’s veterinary drug handbook. 5th ed. Ames: Blackwell; 2005. p. 713-714.
- Rosenberg JM, Nathan JP, Plakogiannis F. Weight variability of pharmacist-dispensed split tablets. J Am Pharm Assoc (Wash) 2002;42(2):200-205.
- Boggie DT, Delattre ML, Schaefer MG, Morreale AP, Plowman BK. Accuracy of splitting unscored valdecoxib tablets. Am J Health-Syst Pharm 2004;61:1482-1483.
- Uniformity of dosage units. United States pharmacopeia 24/national formulary 19: Rockville, MD: United States Pharmacopeial Convention; 1999. p. 2000-2003.
- Rindone JP. Evaluation of tablet-splitting in patients taking lisinopril for hypertension. J Clin Outcomes Manag 2000;7: 22-24.
- Parra D, Beckey NP, Raval HS, Schnacky KR, Calabrese V, Coakley RW, Goodhope RC. Effect of splitting simvastatin tablets for control of low-density lipoprotein cholesterol. Am J Cardiol 2005;95:1481-1483.
- Gee M, Hasson NK, Hahn T, Ryono R. Effects of a tabletsplitting program in patients taking HMB-CoA reductase inhibitors: analysis of clinical effects, patient satisfaction, compliance, and cost avoidance. J Manag Care Pharm 2002; 8(6):453-458.
- Peek BT, Al-Achi A, Coombs SJ. Accuracy of tablet splitting by elderly patients. JAMA 2002;288(4):451-452.
- [No authors listed] Tablet splitting. Med Lett Drugs Ther 2004;46(1195):89-91.
- Mc Devitt JT, Gurst AH, Chen Y. Accuracy of tablet splitting. Pharmacotherapy 1998;18(1):193-197.
- Gupta P, Gupta K. Broken tablets: does the sum of the parts equal the whole? Am J Hosp Pharm 1998;45: 1498.
- Veronin MA, Youan B. Magic bullet gone astray: medications and the internet. Science 2004;305:481.
- Injac R, Djordjevic-Milic V, Srdjenovic B. Thermostability testing and degradation profiles of doxycycline in bulk, tablets, and capsules by HPLC. J Chromatogr Sci 2007;45(9): 623-628.
- Marriott JL, Nation RL. Splitting tablets. Aust Prescr 2002; 25:133-135.
^Наверх









